Week 1: Botanical Explorations with (and without) Dissecting Microscopes
1/11/2016
Goals
Our initial goal was to familiarize ourselves with the dissecting scopes with an emphasis on the usefulness, applications, and skills. We then successfully established a comfortable relationship between ourselves and developed a safe and friendly atmosphere for cooperative learning. The final aspect of our goal was to openly ask questions and prepare our minds for the interestingly awesome botanical journey we have ahead of us.
Our initial goal was to familiarize ourselves with the dissecting scopes with an emphasis on the usefulness, applications, and skills. We then successfully established a comfortable relationship between ourselves and developed a safe and friendly atmosphere for cooperative learning. The final aspect of our goal was to openly ask questions and prepare our minds for the interestingly awesome botanical journey we have ahead of us.
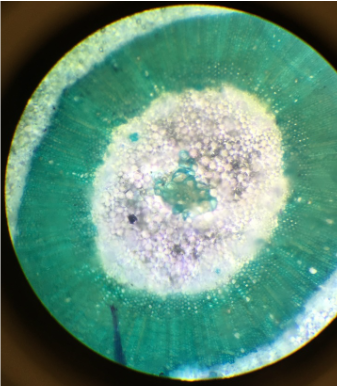
This was our first lab of the semester! We went around campus in search of a plant item apiece. Kevin and I decided to look around the Student Recreation Center, wherein we found our two specimens. We examined each under the dissecting scope and using our unaided eyes (aided in my case). I was most surprised to see such fine hairs on the underside of my leaf, however, I could not feel them.
- Loye
- Loye
Week 2: Plant Cells & Scientific Drawings
1/25/2016
Goals
Our goal for this exercise was to practice preparing a wet-mount slide of plant cells in order for viewing under a compound microscope. We had to recognize the various and different slide preparation techniques of the whole mount, the epidermal peel, the sections (such as longitudinal and cross), as well as smears. Once we achieved recognition we were able to find and identify the visible characteristics of a living plant cell such as the cell wall, vacuole, nucleus, chloroplasts, and the cytoplasm. After we were able to identify the characteristics of a living plant cell we used biological stains in order to make those characteristics visible for a microscopic study. Our final goal was to be able to accurately produce a scientific diagram with a fully labeled living plant cell.
Our goal for this exercise was to practice preparing a wet-mount slide of plant cells in order for viewing under a compound microscope. We had to recognize the various and different slide preparation techniques of the whole mount, the epidermal peel, the sections (such as longitudinal and cross), as well as smears. Once we achieved recognition we were able to find and identify the visible characteristics of a living plant cell such as the cell wall, vacuole, nucleus, chloroplasts, and the cytoplasm. After we were able to identify the characteristics of a living plant cell we used biological stains in order to make those characteristics visible for a microscopic study. Our final goal was to be able to accurately produce a scientific diagram with a fully labeled living plant cell.
Today we reviewed the details and tricks of our compound microscopes, which at the beginning of any semester is always welcomed. We also reviewed how to make wet-mount slides for specimens that are not of the bacterial persuasion. Once we had the basics down we prepared numerous slides to identify various features of a living plant cell. Also, we used two stains, Iodine and Toluidine Blue to help provide contrast so that we could easily view these structures. In addition to all of this we worked on our first scientific diagrams and hashed out the finer details of maintaining our lab notebooks.
- Loye & Kevin (Team Beard)
- Loye & Kevin (Team Beard)
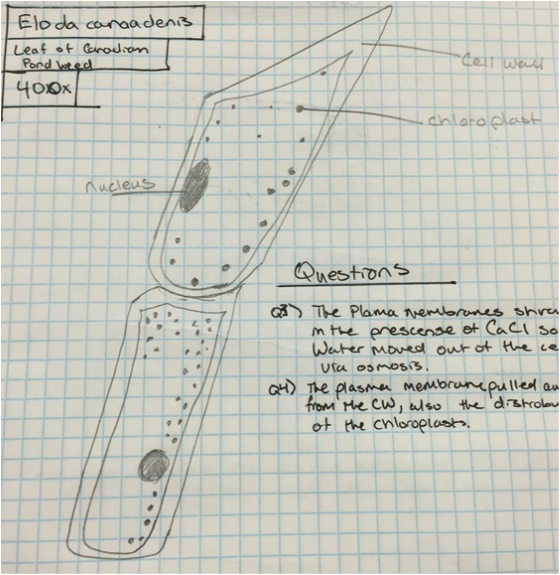
|
In the image to the direct left you can visualize how much the cytoplasm has pulled away from the cell wall. This is due the CaCl solution pulling water from inside the cell and into the environment by osmosis. |
|
In this view we can see the size comparison from each stage of the pepper as it turns read the chromoplasts are larger. The shapes differ as well from round to a more jagged edge in the red pepper. |
|
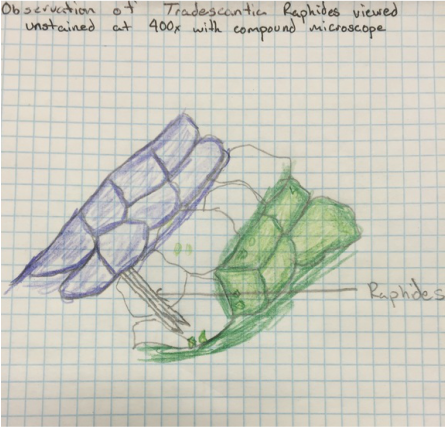
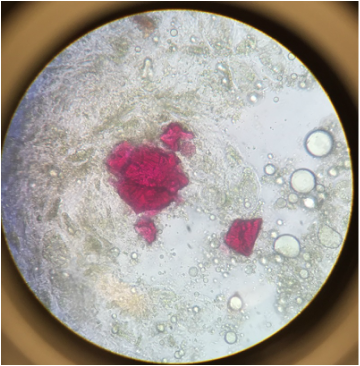
This was my favorite specimen of the day, the Raphides are needle-shaped calcium oxalate crystals to dissuade the eating of this plant. So I like that crystals aren't only formed deep inside of our planet but inside plants as well. - Kevin |
Week 3: Photosynthesis
02/01/2016
Goals
Our goal this week is using chromatography to separate and identify various pigments found in photosynthetic plant tissues. Then develop an explanation of why having more types of photosynthetic pigment would enhance a plant's success. Using an experiment to demonstrate that carbon fixation during the "synthesis" reactions of photosynthesis is dependent on products of the "photo" reactions. Finally using a starch test to examine the effects of carbon-starvation and light-deprivation have on the synthesis reactions of photosynthesis.
- Loye & Kevin (Team Beard)
Our goal this week is using chromatography to separate and identify various pigments found in photosynthetic plant tissues. Then develop an explanation of why having more types of photosynthetic pigment would enhance a plant's success. Using an experiment to demonstrate that carbon fixation during the "synthesis" reactions of photosynthesis is dependent on products of the "photo" reactions. Finally using a starch test to examine the effects of carbon-starvation and light-deprivation have on the synthesis reactions of photosynthesis.
- Loye & Kevin (Team Beard)
|
Tubes 1 and 3 were positioned under lamps, while tubes 2 and 4 were kept in the dark. We used phenol red in water as a pH indicator solution, which at close to neutral is red. We introduced CO2 through a straw, which was held in solution as carbonic acid, changing the solution to a light orange color. After this a sprig of Elodea was placed in the tubes 1 and 4, then respectively left in the light and dark. Tube 1 showed us that the Elodea was still undergoing photosynthesis and raised the pH of the solution, changing the color to red.
Seeing the active science behind photosynthesis was my favorite part of today. Seeing is believing.
- Loye |
|
In the image to the direct left the lighter areas show that starch was present in the locations of the leaf that were green or contained chlorophyll. This was my favorite activity of the day, boiling the leaf and then seeing the difference from staining was science in motion. - Kevin |
Team Beard Sciencing the ---- out of it.
Week 4: Simple and Complex Tissues
2/8/2016
Goals
This week we were to compare and contrast parenchyma, collenchyma, and sclerenchyma cells and tissues, which are part of the ground tissue system. Also, we recognized the three tissue systems of the plant body, dermal, ground, and vascular. We identified the water-conducting cells, the vascular tissue system, and their structural features and functions. And upon an epidermal peel we saw the features of the epidermis.
- Loye & Kevin (Team Beard)
This week we were to compare and contrast parenchyma, collenchyma, and sclerenchyma cells and tissues, which are part of the ground tissue system. Also, we recognized the three tissue systems of the plant body, dermal, ground, and vascular. We identified the water-conducting cells, the vascular tissue system, and their structural features and functions. And upon an epidermal peel we saw the features of the epidermis.
- Loye & Kevin (Team Beard)
|
|
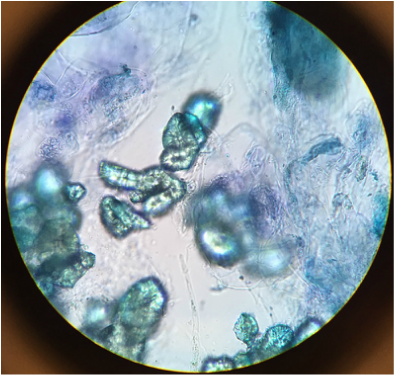
The two tissues that we photographed to the right, and contrasted with Toluidine Blue O, are collenchyma cells and parenchyma cells. The darkly stained areas are the collenchyma cells, stained because of pectins. The lightly, almost translucent cells are the parenchyma tissue, which are precursor cells that later undergo differentiation. |
|
This was my favorite part of the lab. I originally had a section of the snake plant that was not quite what we were looking for. After some confirmation and adjustments from Dr. L-P, we obtained the images seen here. -Loye |
|
|
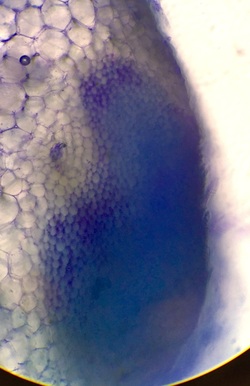
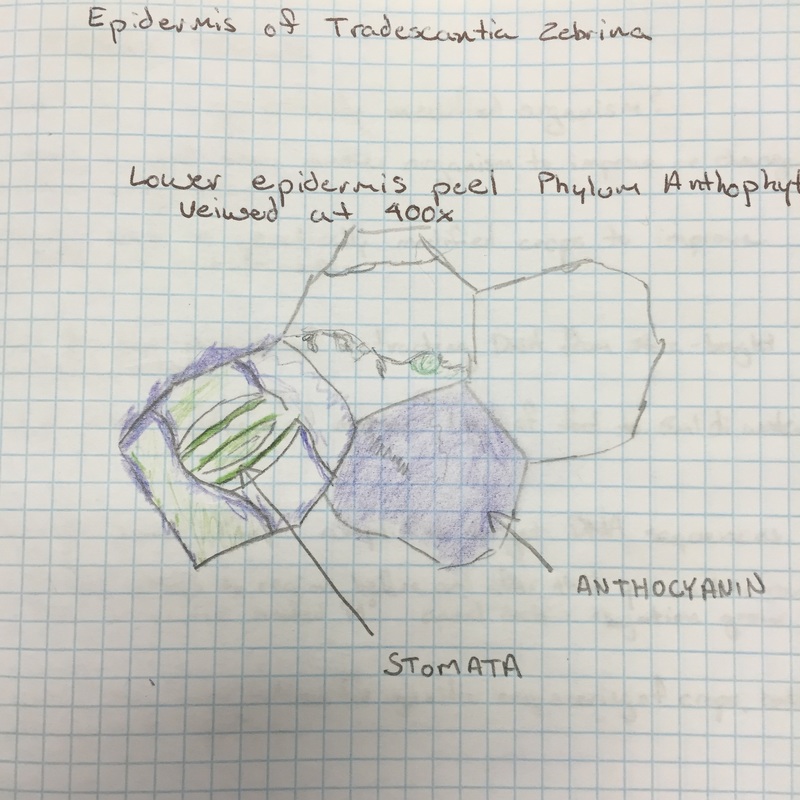
In the image to the right of the epidermal peel of the inch plant (Tradescantia zebrina) there was an absence of chloroplasts through out the peel, with the exception of the guard cells of the stomata. Also, we observed the pigment to the right of the bottom-most stomata and identified it as anthocyanin. |
|
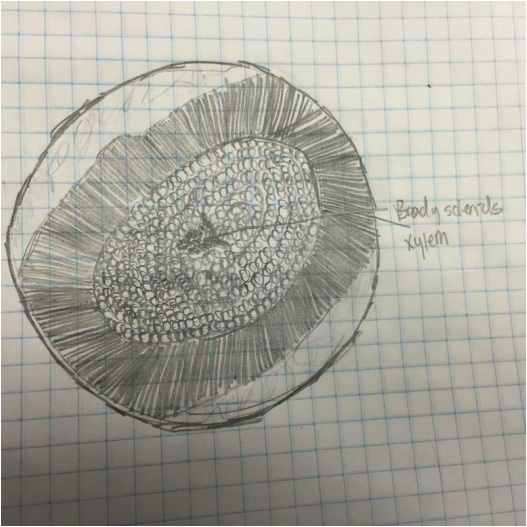
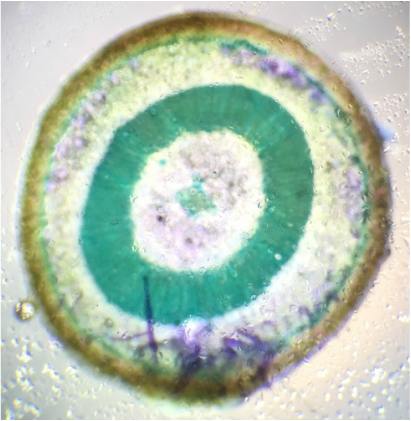
We observed brachysclereids in the pith, positioned directly in center of both images. The surrounding green, lignified ring is the xylem, which mostly transports water. Further out from that are the also green, lignified cells that make up the cortex as seen in the wider cross-section on the left. |
Week 5 : GMO Investigation, Part I
2/22/2016
Goals
This week we learned that organisms that have had particular genes introduced into their genomes are classified as genetically modified. To further understand this concept we went through the process of extracting DNA from common food items. We achieved this by grinding samples into slurry with deionized water to a specified concentration, then we added it to a screwcap tube containing InstaGene matrix. These negatively charged beads allow for the separation of positively charged minerals from the negatively charged DNA to allow it to run through PCR. The PCR achieves the amplification of target DNA sequences by using unique primers for those particular sequences, then it uses existing DNA replication machinery that are borrowed from thermophilic Archaea. Through submitting the samples to various temperatures it forces the DNA to undergo replication on an exponential scale. Also, watched a film in class that showed the massive economical, social, and environmental impact that genetically modified organisms have on our pale blue dot.
- Loye & Kevin (Team Beard)
This week we learned that organisms that have had particular genes introduced into their genomes are classified as genetically modified. To further understand this concept we went through the process of extracting DNA from common food items. We achieved this by grinding samples into slurry with deionized water to a specified concentration, then we added it to a screwcap tube containing InstaGene matrix. These negatively charged beads allow for the separation of positively charged minerals from the negatively charged DNA to allow it to run through PCR. The PCR achieves the amplification of target DNA sequences by using unique primers for those particular sequences, then it uses existing DNA replication machinery that are borrowed from thermophilic Archaea. Through submitting the samples to various temperatures it forces the DNA to undergo replication on an exponential scale. Also, watched a film in class that showed the massive economical, social, and environmental impact that genetically modified organisms have on our pale blue dot.
- Loye & Kevin (Team Beard)
|
We chose to use Cinnamon Toast Crunch (Innamoncay oastay-unchcray) cereal as our test food to determine if it contained any GMO ingredients. Through discussion with Dr. L-P we learned that genetically modified wheat has not been approved for use in the United States. As wheat is the main ingredient in this cereal we hypothesize that it will not show the presence of GMOs. The cereal does contain soy lecithin that is sourced from soybeans, of which a large percentage have been genetically modified.
|
|
The purpose of using the non-GMO raw rolled oats was to provide a negative control to our experiment. When we later run look for the presence of GMOs in the sample containing this we should find none whatsoever, if GMO is present contamination of our samples has occurred. |
|
We created a slurry to mix with the InstaGene matrix that consisted of the food sample Innamoncay oastay-unchcray and deionized water. This allowed for disruption of the tissue allowing for a more complete separation of the DNA from other minerals that would interfere with PCR.
My favorite part about this lab was using the mortar and pedestal, because you really don't get that many opportunities to use them. Also, they were produced by Coors and historically helped them remain in operation during prohibition. - Loye |
|
After creating the slurry it is then placed in a screw cap tube with InstaGene matrix. The sample is then placed in the water bath for 5 minutes. This allows for the aggregation of negatively charged InstaGene beads and positively charged minerals that could assist enzymes to degrade the DNA. The heat also destroys the enzymes. |
|
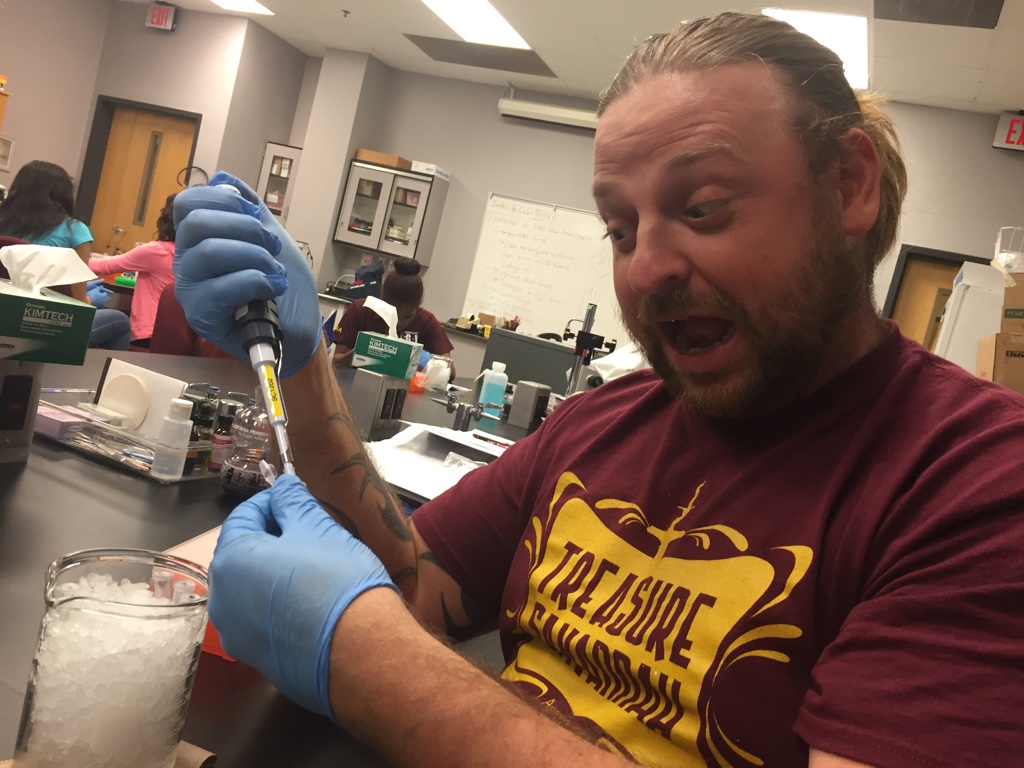
As you can tell from the excitement on my face...yes that is excitement....this is my favorite part of the lab by showing off my mad pipetting skills. - Kevin
|
At this stage we added green plant master mix and red GMO master mix to the PCR tubes. The master mixes allows for the amplification of genes that are specifically found in all plants and GMO, respectively. For the plant master mix it amplifies a section of a chloroplast gene used by photosystem II that should be ubiquitous in all plants. The GMO master mix amplifies DNA sequences that are found in approximately 85% of all GMO plants. The master mixes contain primers for the sequences we wish to amplify as well as Taq DNA polymerase, dNTPs, MgCl2, and buffers. We then added the samples (non-GMO, sample food, and provided GMO positive sample) to the PCR tubes and kept them on ice to prevent degradation.
|
|
The PCR reaction then requires a series of specific temperature fluctuations to allow for optimum replication of the DNA. The first step is to increase the temperature to allow for the denaturation of the DNA from a corkscrew shape to a more linear form. Then it quickly drops the temperature to allow for the annealing of primers to the target areas of the DNA. The last step is to bring the temperature back up, but not quite to the extreme of the first step, allowing for the Taq DNA polymerase to extend the complementary DNA to the template strands. This cycle then repeats 40 times. |
Week 6: GMO Part II The conclusion
2/29/2016
Goals
This week we concluded our GMO investigation by electrophoresis to analyze our samples for presence of GMO primers. We learned the basics of electrophoresis wherein the negatively charged DNA is pulled through the agarose gel to the positive side of the tray. We placed our samples in the wells of the gel slab with a molecular weight ruler to measure results. We then took the slab of gel and placed it in the ChemiDoc, which uses UV light to visualize the PCR products. Using the molecular weight ruler we were able to quantify the length of the primers amplified by the PCR. We were able to visually recognize the importance of the negative and positive controls used to ensure the accuracy of our data and eliminate any contamination. While the electrophoresis was running we watched a film that detailed the history and implications of genetically modified crops. The film illustrated how large of a reach Monsanto has on crop production and how they use the legal system to their advantage while hurting farmers.
- Loye & Kevin (Team Beard)
This week we concluded our GMO investigation by electrophoresis to analyze our samples for presence of GMO primers. We learned the basics of electrophoresis wherein the negatively charged DNA is pulled through the agarose gel to the positive side of the tray. We placed our samples in the wells of the gel slab with a molecular weight ruler to measure results. We then took the slab of gel and placed it in the ChemiDoc, which uses UV light to visualize the PCR products. Using the molecular weight ruler we were able to quantify the length of the primers amplified by the PCR. We were able to visually recognize the importance of the negative and positive controls used to ensure the accuracy of our data and eliminate any contamination. While the electrophoresis was running we watched a film that detailed the history and implications of genetically modified crops. The film illustrated how large of a reach Monsanto has on crop production and how they use the legal system to their advantage while hurting farmers.
- Loye & Kevin (Team Beard)
|
The gel electrophoresis rig has two charged poles next to the samples. The black pole is negatively charged, while the red is positively charged. DNA molecules inherently have a negative charge, which allows them to be pulled through the gel to the positive end of the rig. With the help of a molecular weight marker we are able to analyze the size of the DNA molecules by measuring the amount of base pairs that compose them. |
|
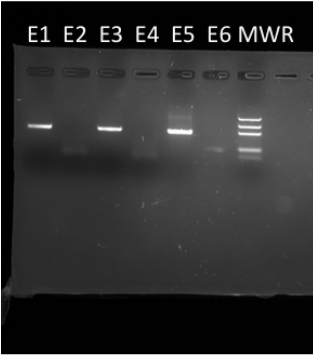
To the right you can visualize the results of our samples after PCR and gel electrophoresis. In lane E1, which consisted of our Non-GMO food sample with our PMM showing a band at approximately 445bp, which acts as the positive control for the Non-GMO food sample. In lane E2, which consisted of our Non-GMO food sample with our GMO MM showing no band, which acts as the negative control for the Non-GMO food sample. In lane E3, which consisted of our food sample of Cinnamon Toast Crunch (Innamoncay oastay-unchcray) with our PMM showing a band at approximately 445bp, showing that we do have DNA extracted from a plant. In lane E4, which consisted of our food sample of Cinnamon Toast Crunch (Innamoncay oastay-unchcray) with our GMO MM showing no band, which infers that there was no genetically modified DNA in our food sample. In lane E5, which consisted of our GMO food sample with our PMM showing a band at approximately 445bp, which acts as the negative control for the GMO food sample. In lane E6, which consisted of our GMO food sample with our GMO MM showing a band at approximately 200bp, which acts as the positive control for the GMO food sample. Lastly, in lane seven, the furthest to the right, is our Molecular Weight Rule allowing us to quantify our data in units of base pairs.
|
Results
Our experiments showed that our sample food, Cinnamon Toast Crunch (Innamoncay oastay-unchcray), did not contain genetically modified DNA due to the lack of any bands appearing in lane E4. By using our controls in lanes E1, 2, 5, and 6, we can state with confidence that our results were accurate and precise. However, our lab section did encounter an anomaly in lane E6, wherein we visualized a band at 445bp in the GMO food sample with GMO MM. This could be due to contamination from preparing samples or potentially a bad batch of master mix. If we were to repeat this procedure we would focus on ensuring absolutely no contamination during preparation of samples and use more skill with pipetting our samples in the gel electrophoresis slab wells.
Our experiments showed that our sample food, Cinnamon Toast Crunch (Innamoncay oastay-unchcray), did not contain genetically modified DNA due to the lack of any bands appearing in lane E4. By using our controls in lanes E1, 2, 5, and 6, we can state with confidence that our results were accurate and precise. However, our lab section did encounter an anomaly in lane E6, wherein we visualized a band at 445bp in the GMO food sample with GMO MM. This could be due to contamination from preparing samples or potentially a bad batch of master mix. If we were to repeat this procedure we would focus on ensuring absolutely no contamination during preparation of samples and use more skill with pipetting our samples in the gel electrophoresis slab wells.
Our heroes did not find the GMO baddies this week but kids stay ever vigilant on what you EAT
Week 7: Bryophytes & Ferns
Goals
This is our first lab back from Spring Break, so we had to get back into the swing of things. We examined members of the Phylums Bryophyta and Hepatiocphyta, which were represented by mosses and liverworts respectively. Also, we were able to use the nomenclature associate with the two nonvascular taxons. We then examined some Monilophytes taking notice of their macroscopic structural features as well as their microscopic features, such as lignified tracheids. We also expanded on our knowledge of Alterations of Generations by identifying the gametophyte and sporophyte generations and their predominance in the varying species.
This is our first lab back from Spring Break, so we had to get back into the swing of things. We examined members of the Phylums Bryophyta and Hepatiocphyta, which were represented by mosses and liverworts respectively. Also, we were able to use the nomenclature associate with the two nonvascular taxons. We then examined some Monilophytes taking notice of their macroscopic structural features as well as their microscopic features, such as lignified tracheids. We also expanded on our knowledge of Alterations of Generations by identifying the gametophyte and sporophyte generations and their predominance in the varying species.
|
The image to the right of the Sphagnum gametophyte shows no change from the image of the same specimen on the left. The image on the right has been stained with Phloroglucinol HCl, which would only stain to pectin and lignin, of which there is none in this plant. Therefore we have verified that our plant has no lignin. |
This was my favorite activity of lab this week. I really enjoy going out and getting plants to look at in lab that are found on campus. - Kevin
|
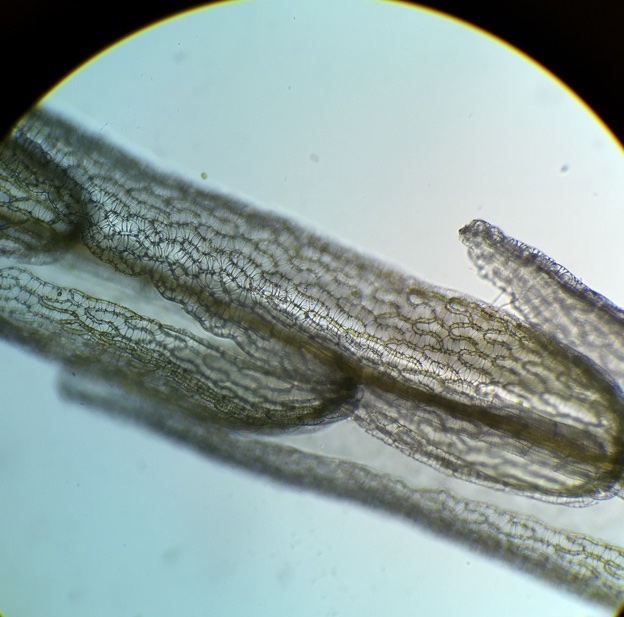
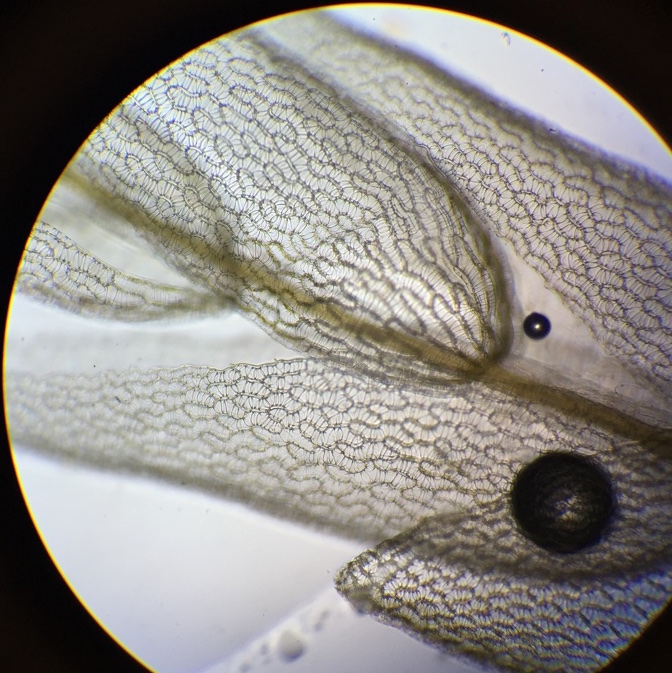
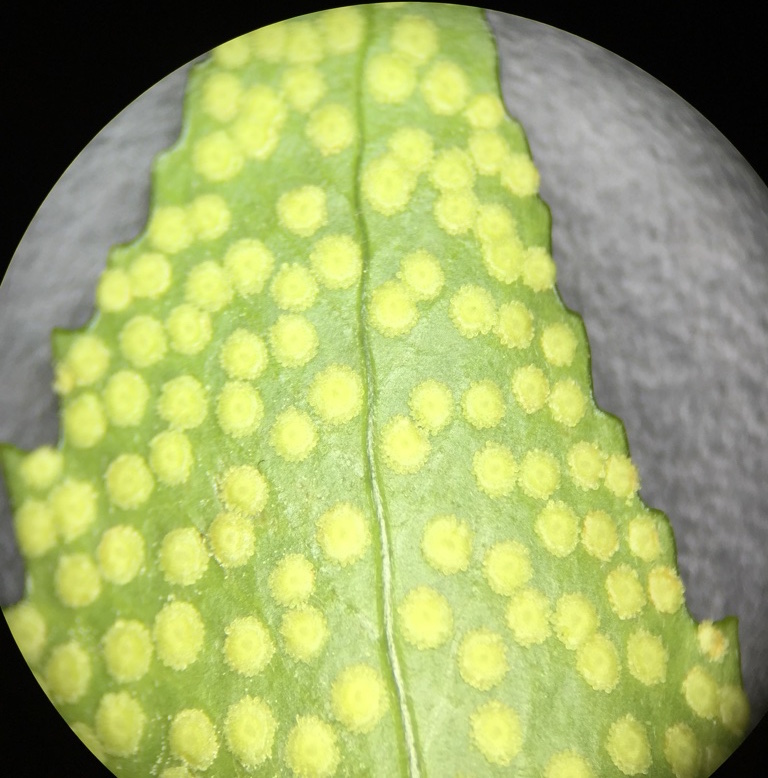
In the photo to the left you can see yellow dots on the abaxial surface of a fern frond, which are called sori. They are clusters of sporangia covered by indusim that provide protection until the spores mature. Within the sporangia spores are produced via meiosis, which when released are hermaphroditic in nature. The spores then give rise to the gametophytes. |
|
In the image to the right you can see the red stained, lignified tracheary elements of the Psilotum. There is also a hint of lignin to the far right of the image as well, however when preparing the slide, I cut my cross-section with a slight angle. |
|
This was my favorite part of the lab, just watching the sperm swim around and collide with one another. - Loye That would be your favorite part I shouldn't say this I might be sori. - Kevin |
Week 8: Gymnosperms & Angiosperms
Seed Plants
Goals
This week we move forward in the evolution of plants with the introduction of the Seed Plants: Gymnosperms and Angiosperms. By the end of lab we should be able to recognize plants from both taxon, as well as describe the different sequences of events for the gametophyte and sporophyte generations. In addition we will beobserving flowers and describing their functions using the terms ovary position, presence/absence of floral organs, and floral symmetry. We will follow up our trip around campus with a floral dissection of a specimen that chose in the wilderness that is Armstrong State University.
This week we move forward in the evolution of plants with the introduction of the Seed Plants: Gymnosperms and Angiosperms. By the end of lab we should be able to recognize plants from both taxon, as well as describe the different sequences of events for the gametophyte and sporophyte generations. In addition we will beobserving flowers and describing their functions using the terms ovary position, presence/absence of floral organs, and floral symmetry. We will follow up our trip around campus with a floral dissection of a specimen that chose in the wilderness that is Armstrong State University.
|
|
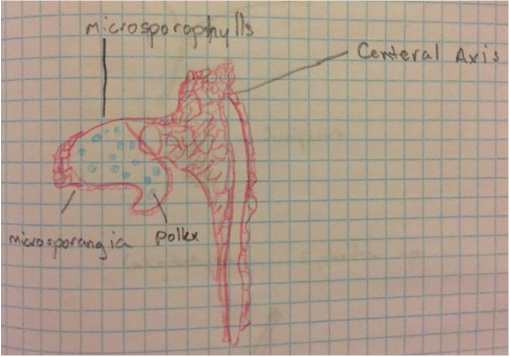
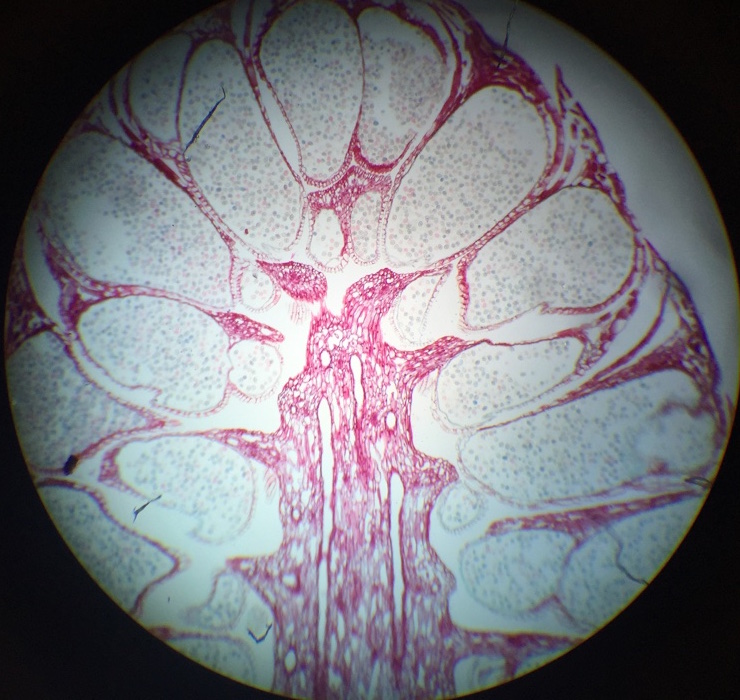
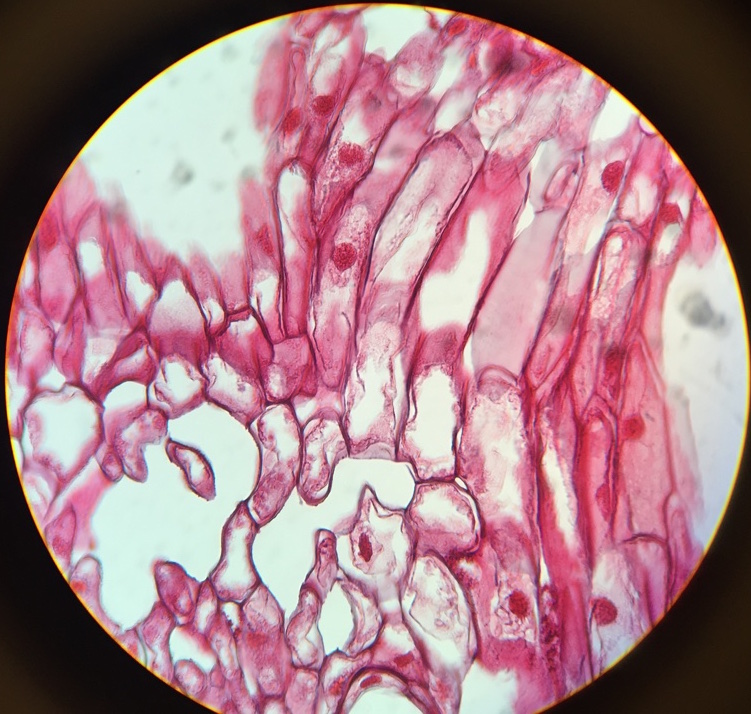
Seeds are formed on the upper side of the cone scale in female cones, towards the center of the cone. |
|
This Ginkgo biloba specimen is a male, because the seeds produced by the female plants have a “rancid butter” smell, therefore they are not popular to have around where people walk. Also, the leaves of this tree are deciduous, meaning that they change color and drop once a year. |
Loye's Flower Dissection
Kevin's Flower Dissection
This was a hard choice this week I have to say that I had two favorites this dissection and the walk around campus. It was great seeing all the trees and flowers that I have walked by over the years up close. -Kevin
Week 9: Getting to the Root of Roots
4/11/16
Goals
We observed the different methods of root grow between the monocots and dicots, wherein the dicot's primary root is the taproot and the monocots create short-lived primary roots, eventually developing into a fibrous root system. To better understand the three regions of morphology for roots, we observed the Region of Cell Division where the root produces new growth via mitosis. We were also able to observe parts of the Region of Elongation, wherein the cells elongate, creating longitudinal growth of the root. Lastly, we observed the Region of Cell Maturation, which differs between dicots and monocots. The monocot root has a central area that spreads the xylem in a circular pattern around it, whereas the dicots lack this allowing the xylem to remain central in the root. We then took our C-Fern cultures, that we started on 3/7/16, and observed the roots, the formation of the sporophyte generation, and finally transplanted our cultures to soil!
We observed the different methods of root grow between the monocots and dicots, wherein the dicot's primary root is the taproot and the monocots create short-lived primary roots, eventually developing into a fibrous root system. To better understand the three regions of morphology for roots, we observed the Region of Cell Division where the root produces new growth via mitosis. We were also able to observe parts of the Region of Elongation, wherein the cells elongate, creating longitudinal growth of the root. Lastly, we observed the Region of Cell Maturation, which differs between dicots and monocots. The monocot root has a central area that spreads the xylem in a circular pattern around it, whereas the dicots lack this allowing the xylem to remain central in the root. We then took our C-Fern cultures, that we started on 3/7/16, and observed the roots, the formation of the sporophyte generation, and finally transplanted our cultures to soil!
|
In the image to the right you can see the fine root hairs extending out from the root from the Region of Maturation. At the end of the root you can see the root cap, which provides protection to the apical meristem and is part of the Region of Division. |
|
In the image to the left we have used the eyepiece pointer to identify a cell undergoing mitosis. The cell is currently undergoing the anaphase stage, wherein the chromatids are being pulled to opposite ends of the cell. Also, to the left of the eyepiece pointer and circled in red, we identified another cell undergoing the same phase of mitosis. |