Christine Agosto, Michelle Shane, & Bryana Rounds
Botanical Explorations with the Dissecting Scopes, Week 1, 1/11/16
Objectives:
Objectives:
- familiarize students with usefulness and applications of the dissecting scopes
- develop student skills in use of dissecting scopes
- stimulate curiosity for botanical study and encourage questions
- establish relationships between students and develop safe atmosphere for cooperative learning
Plant Cells and Scientific Drawings
January 25,2016
The purpose of this lab was to practice making wet slides to be studied with a compound microscope. Wet slides were created using the epidermis of Allium cepa (onion) cells; Elodea canadensis (Canadian Pondweed) leaves; Capsicum annuum (bell pepper) fruit; Tradescantia (spiderwort) stem; Solanum tuberousum (potato) parenchyma as well as the parenchyma of the banana fruit.
Different techniques were used to help differentiate structures in each cellular wet mount. Toluidine blue was used to illuminate the cell wall and nucleus of the allium cepa cells. Calcium chloride was added to the elodea canadensis to demonstrate osmosis of water into the cells, thus condensing the chloroplasts visible under the compound microscope as well as illuminating cytostreaming in the cell. The plastids of red, yellow and green capsicum annum fruit were compared at 400x total magnification to delineate the size difference of each plastid type amongst the three peppers. The stamen hairs of the tradescantia plant were studied utilizing the compound microscope after a wet slide was prepared. Special attention was paid to the anthocyanin pigment found in the tradescantia cells which indicated the location of vacuoles to the viewer. Wet mounts from unripened and over-ripened banana fields were prepared with iodine to show the difference in the amount of starch available in the ripened banana versus the unripened banana; the unripened banana was found to contain more starch than it's ripened counterpart. Lastly, the solanum tubersoum plants were also treated with iodine to help detect the presence of amyloplasts within the cell.
Different techniques were used to help differentiate structures in each cellular wet mount. Toluidine blue was used to illuminate the cell wall and nucleus of the allium cepa cells. Calcium chloride was added to the elodea canadensis to demonstrate osmosis of water into the cells, thus condensing the chloroplasts visible under the compound microscope as well as illuminating cytostreaming in the cell. The plastids of red, yellow and green capsicum annum fruit were compared at 400x total magnification to delineate the size difference of each plastid type amongst the three peppers. The stamen hairs of the tradescantia plant were studied utilizing the compound microscope after a wet slide was prepared. Special attention was paid to the anthocyanin pigment found in the tradescantia cells which indicated the location of vacuoles to the viewer. Wet mounts from unripened and over-ripened banana fields were prepared with iodine to show the difference in the amount of starch available in the ripened banana versus the unripened banana; the unripened banana was found to contain more starch than it's ripened counterpart. Lastly, the solanum tubersoum plants were also treated with iodine to help detect the presence of amyloplasts within the cell.
Exercise 2a: Epidermis of Allium Cepa (onion) cells
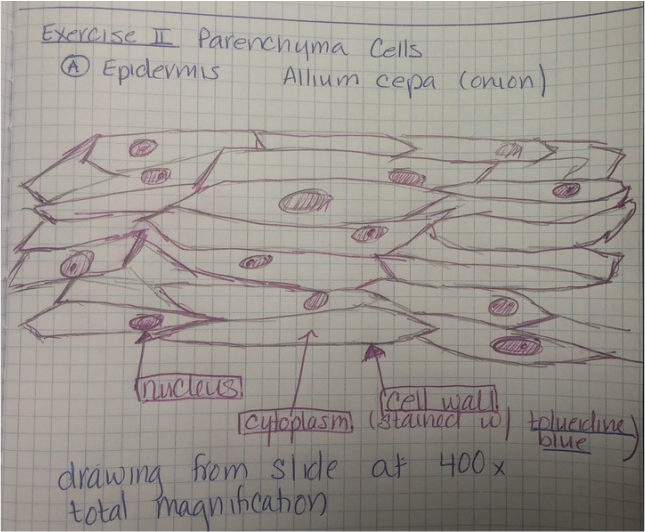
Image 2. Photograph taken of sketch made of the Allium cepa after being stained with toluidine blue in Michelle Shane's lab notebook.
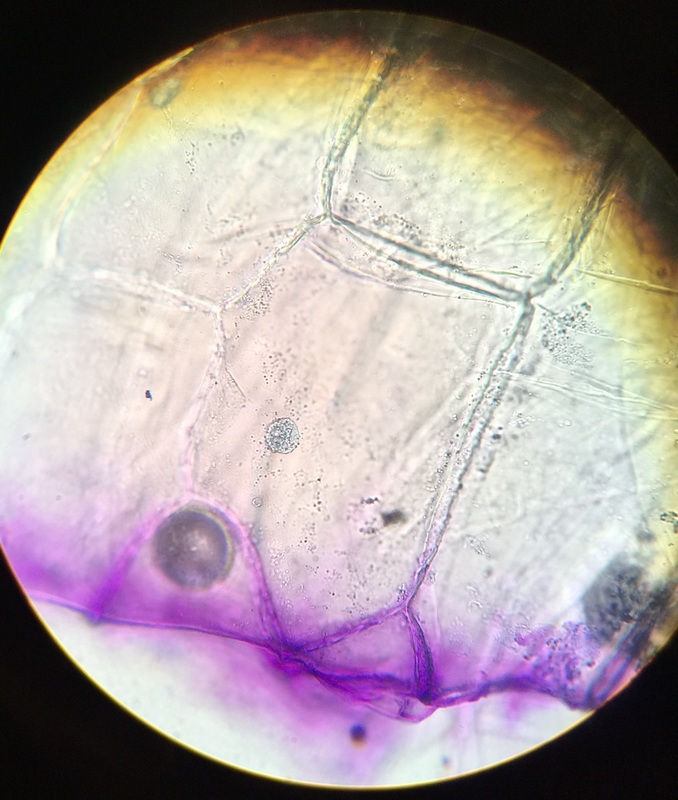
The nucleus, cytoplasm and cell wall are noted in the diagram. The total magnification of the compound microscope was 400x.
The nucleus, cytoplasm and cell wall are noted in the diagram. The total magnification of the compound microscope was 400x.
Exercise 2b: Observation of Canadian Pondweed (elodea canadensis)
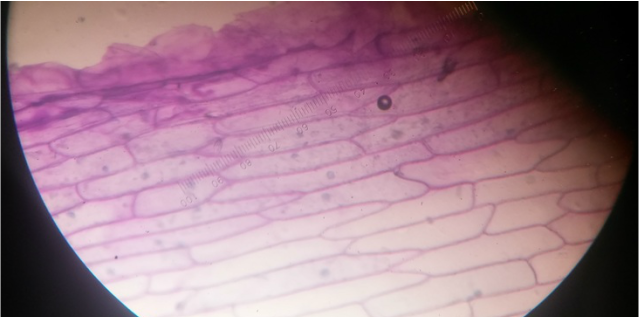
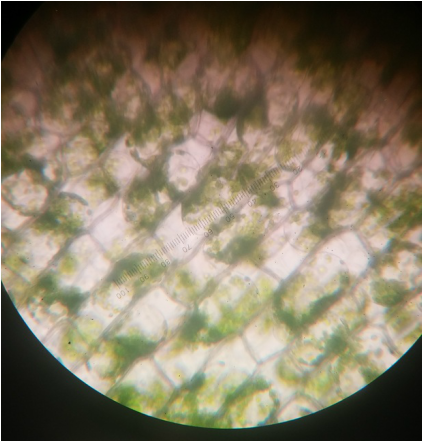
Elodea canadensis prior to the addition of calcium chloride (CaCl). Photographed at 400x total magnification, the chloroplasts appear evenly distributed amongst the cells. The dark line in the dissecting the specimen is the vascular tissue of the leaf. - Michelle
Elodea canadensis at 400x total magnification. Cytoplasmic streaming was observed at this magnification as CaCl was added to the wet mount. It also appears that the once evenly distributed chloroplasts have migrated to the edges of the cell, most likely due to osmotic movement of water into the cells.
-Michelle
-Michelle
Exercise 2c: Observation of Plastids in Cells of Bell Pepper (Capsicum annum)
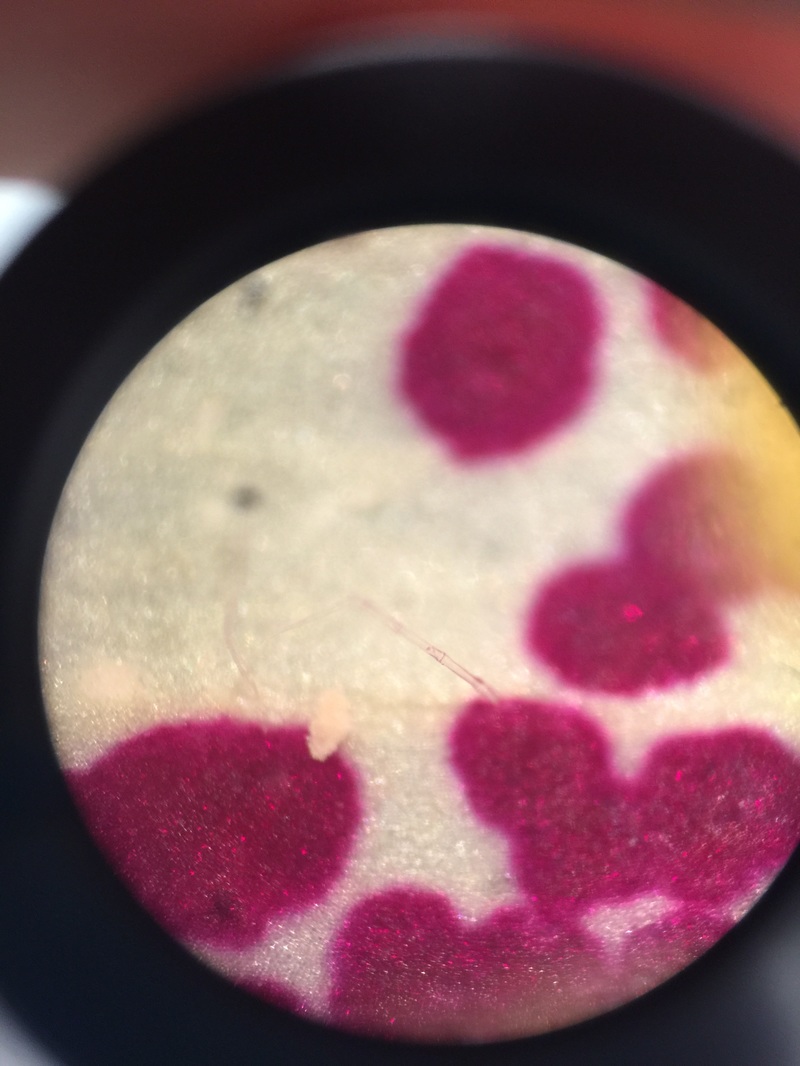
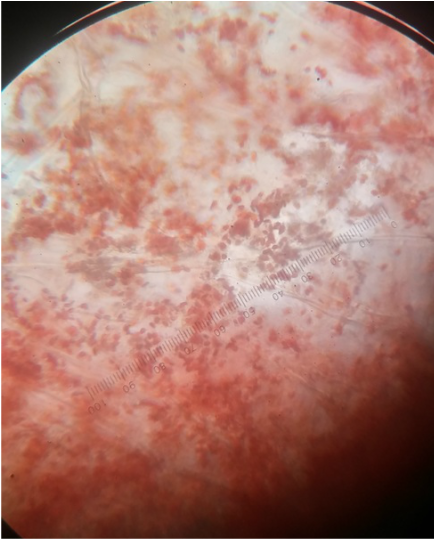
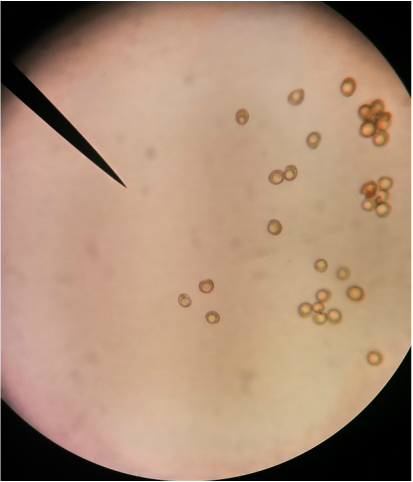
Photograph of a red capsicum annuum (bell pepper) while magnified at 400x total magnification using a oil-immersion objective. The chromoplasts of the cell appear as red dots amongst the cells and were found to be the largest chromoplasts between the three peppers studied.-Michelle
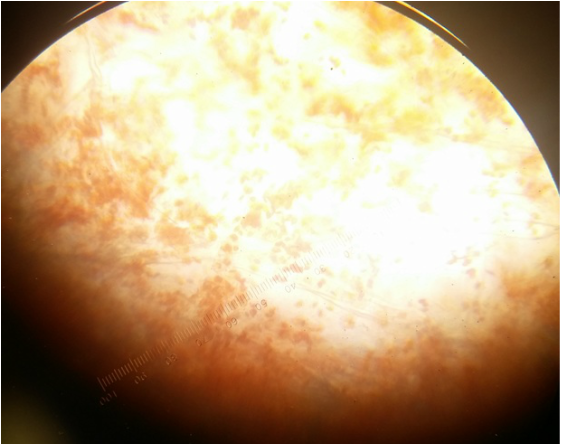
Plastids of the yellow capsicum annum under 400x total magnification. An oil-immersion objective was used to capture details that a 40x dry objective can't. The chromoplasts of the yellow bell pepper are smaller than the chromoplasts of the red bell pepper-Michelle
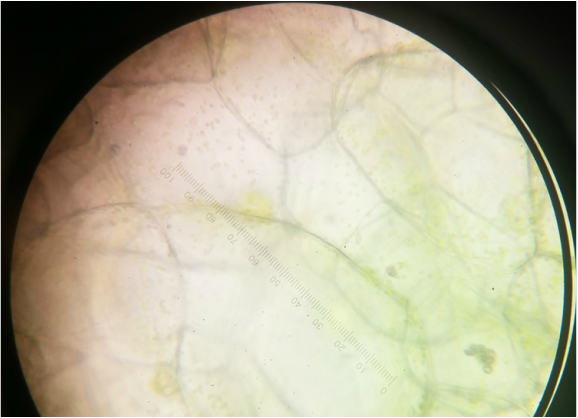
Image of a green capsicum annum under 400x magnification using an oil-immersion objective. The amount of chloroplast appears to be less in comparison to the number of plastids in the red and yellow capsicum annuum. They also appear to be the smallest in size. - Michelle
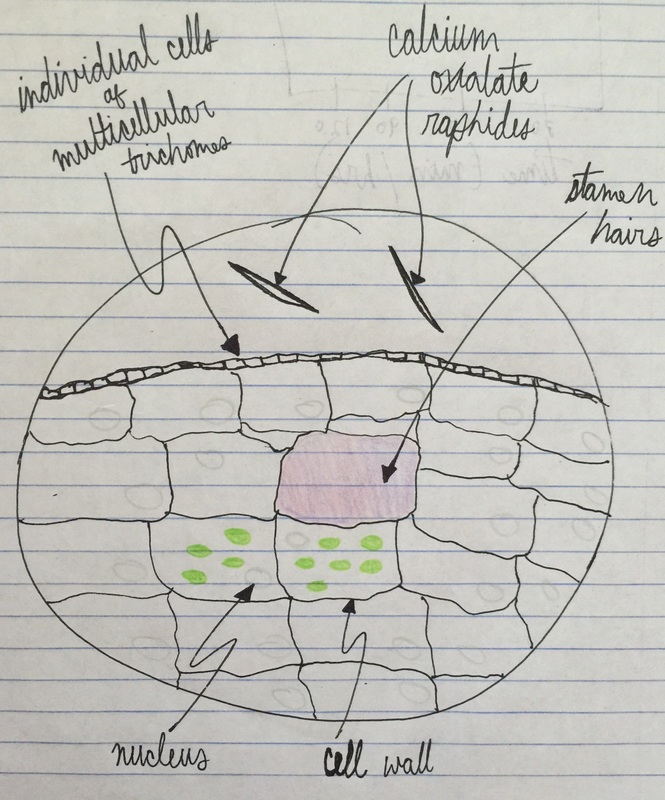
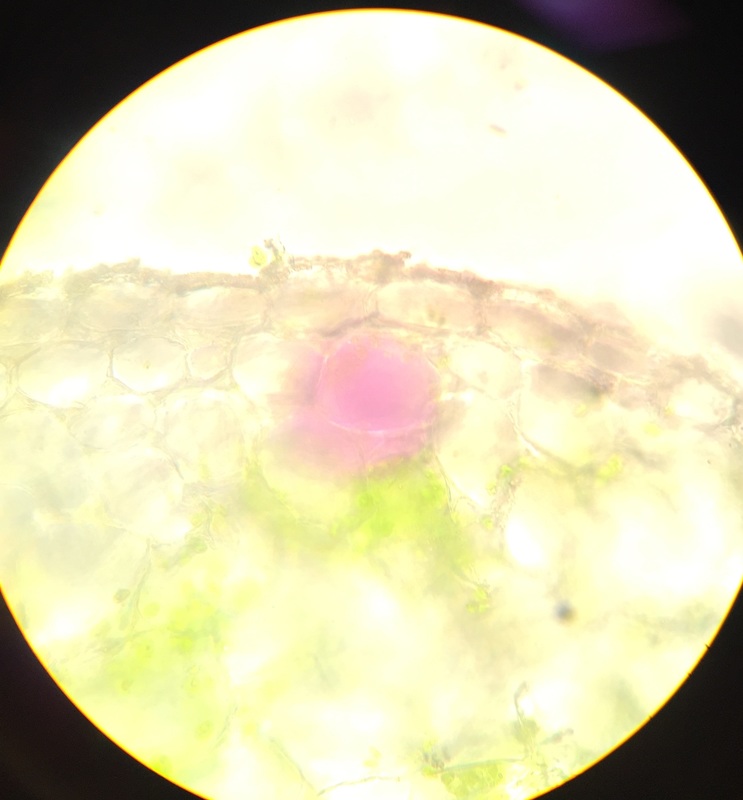
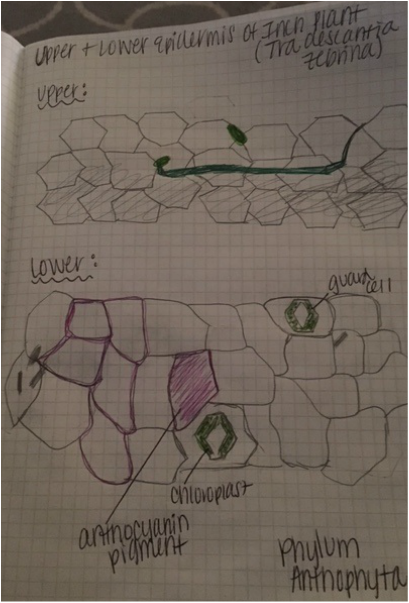
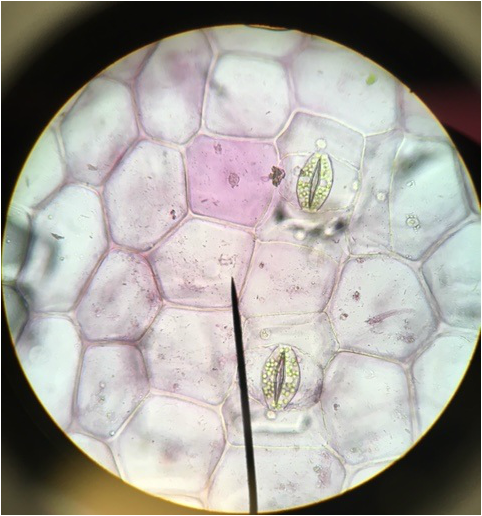
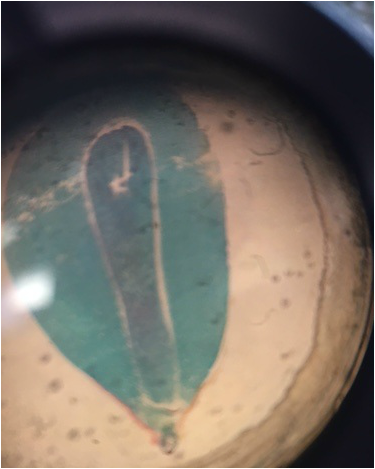
Image shows drawing of Tradescantia cells. The lavender color comes from anthocyanin pigments contained within the vacuoles. The needle-like structures are calcium oxalate raphides. The nucleus is shown in gray, the chloroplasts in green, and the cell wall is represented by the black outlining that defines the cells' limitations. Drawing made from 400x total magnification viewing of wet mounted cross section of Tradescantia, compound microscope. - Christine
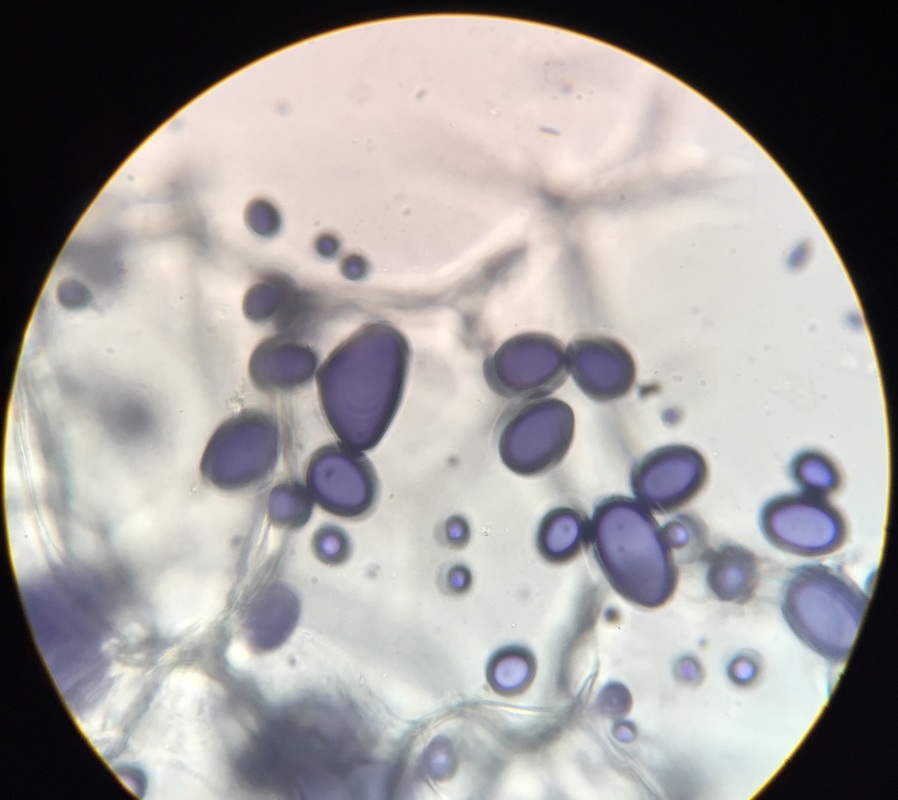
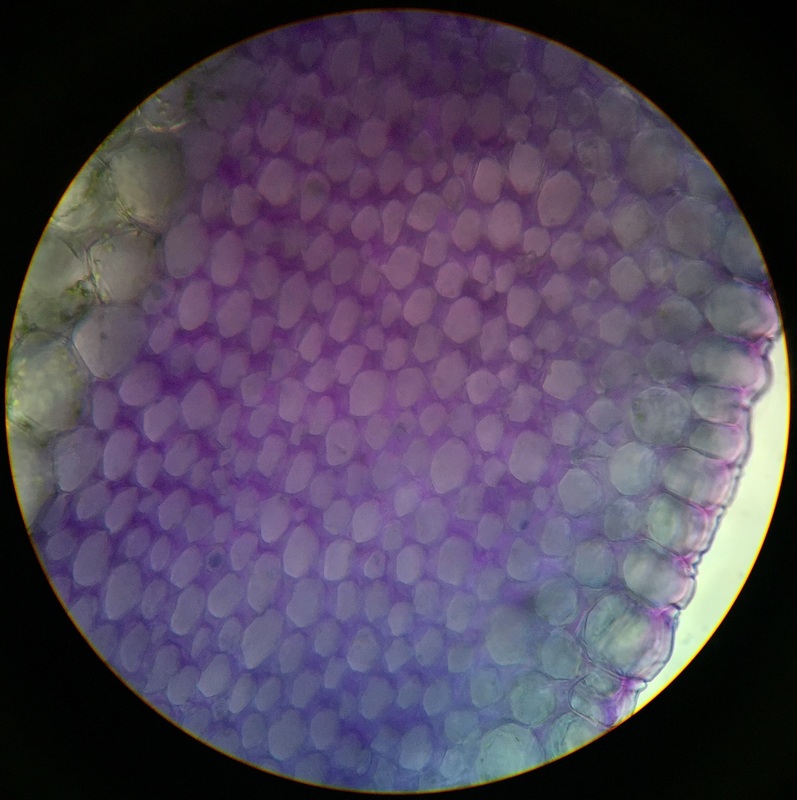
Image shows the cell walls and amyloplasts of Solanum tuberosum (potato). Iodine staining was used. The cell walls appear as dark outlines defining the boundaries of the cell. The amyloplasts (stained dark purple) can be seen inside the cells. Photo taken at 400x total magnification, compound microscope. Sample was prepared by first radial longitudinal sectioning and then cross sectioning of that piece. Section was then wet mounted. - Christine
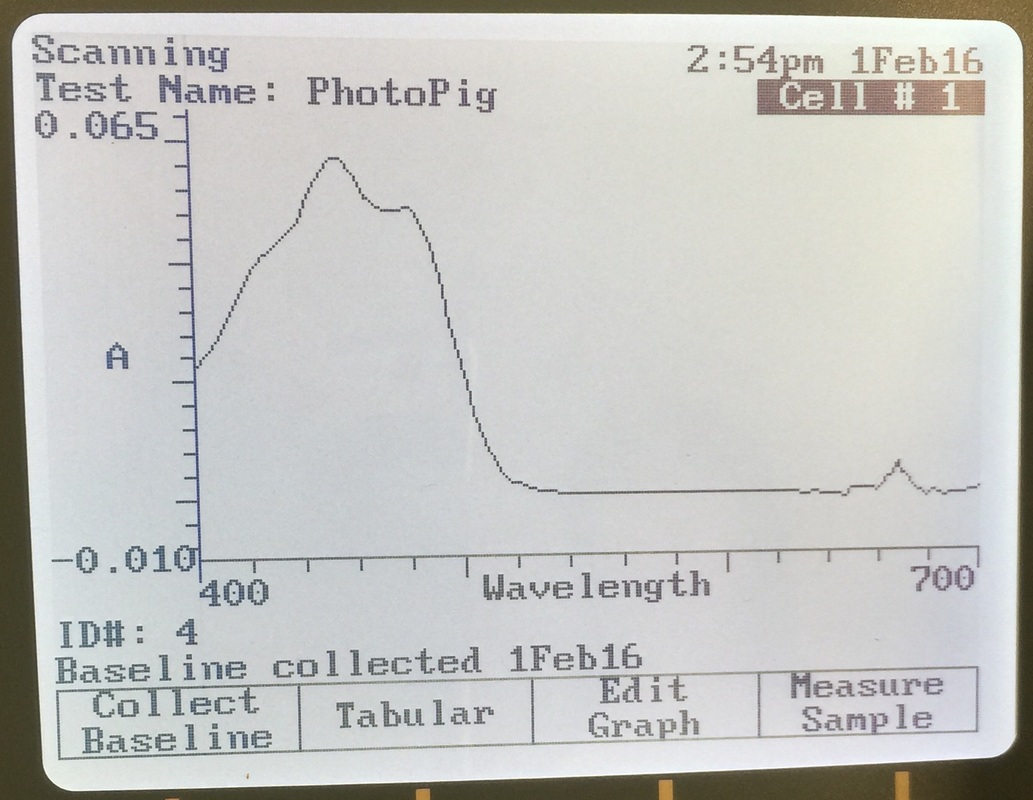
Photosynthesis, Week 3, 2/1/16
Objectives:
- use chromatography to separate and identify various pigments found in photosynthetic plant tissues.
- explain why it would enhance a plant’s success to have more than one type of photosynthetic pigment.
- demonstrate that carbon fixation during the “synthesis” (light-independent) reactions of photosynthesis is dependent on products of the light-dependent reactions (that is, no carbon will be synthesized into carbohydrates unless the light-dependent reactions are taking place simultaneously).
- use a starch test to examine the effects that carbon-starvation and light-deprivation have on the synthesis reactions of photosynthesis.
Simple and Complex Tissues, Week 4, 2/8/16
In today's lab, using different sectioning techniques, we stained various cells and viewed them under the microscope.
Objectives:
Objectives:
- compare and contrast parenchyma, collenchyma, and sclerenchyma cells and tissues (ground tissue system).
- recognize the three tissue systems of the plant body.
- identify water-conducting cells of the vascular tissue system and relate their structural features with their functions.
- describe the characteristics of the epidermis.
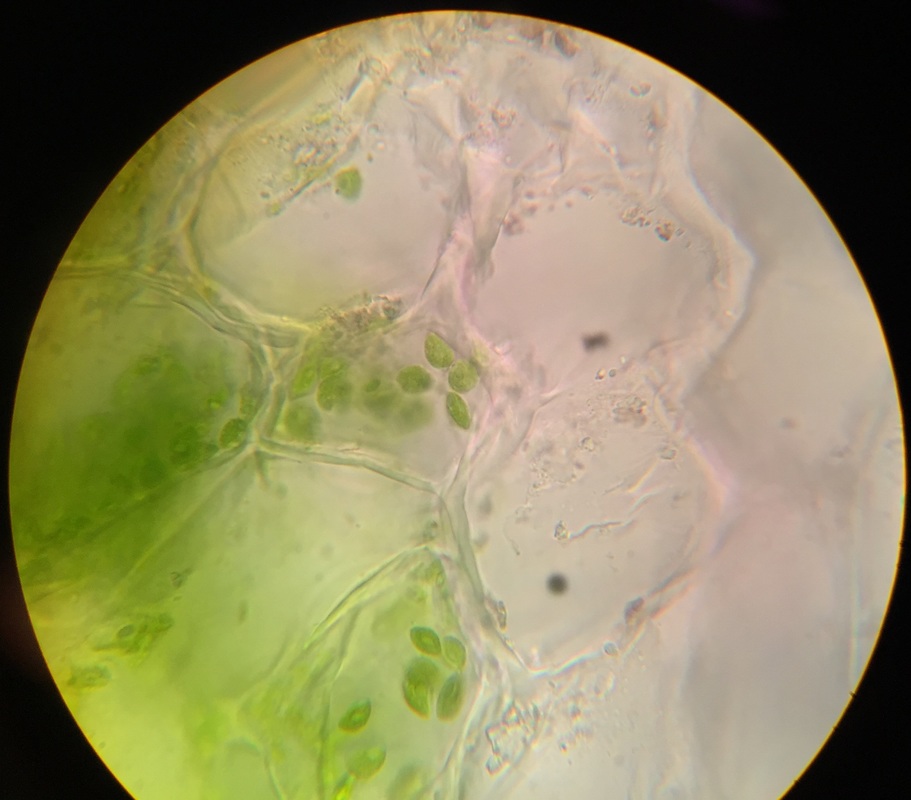
Sclerenchyma of pear fruit, Pyrus communis, stained with TBO. Specimen was obtained by gently scraping some of the pear flesh and smearing it onto the slide. Sclerenchyma cells have thick, lignified cell walls. Brachysclereids, or stone cells, are responsible for the gritty texture of pears. Sclerids are found as single cells or in small groups in the parenchyma tissues. Image taken at 400x total magnification under the compound microscope. – Christine
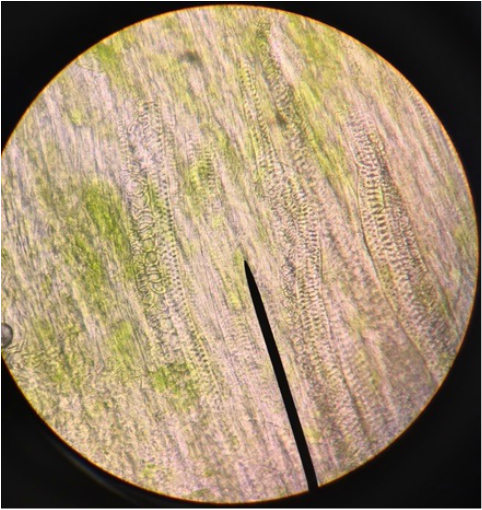
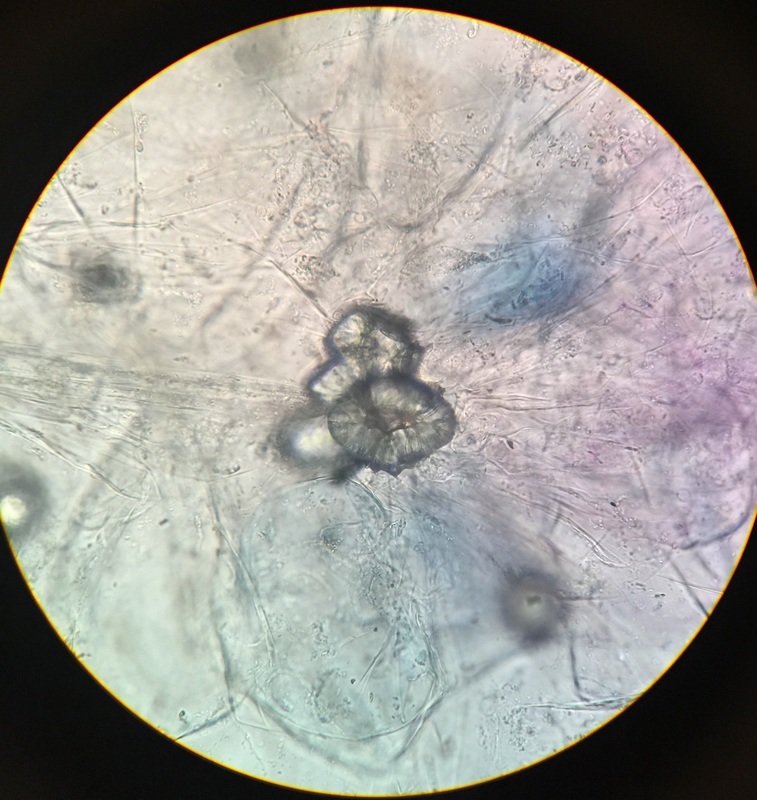
Sclerenchyma fibers of snake plant, Sansevieria trifasciata, obtained by doing a cross-section and stained with TBO. Sclerenchyma fibers provide support to the elongated leaves of the plant. They appear very small (thin) and are stained bright blue because of the lignin in their thick secondary cell walls. Surrounding cells are those of the mesophyll; their cell walls have stained purple due to the presence of cellulose. The vacuoles are clearly visible in these cells and chloroplast are seen around the vacuole indicating that cytoplasmic streaming is occurring. Image taken at 400x total magnification under the compound microscope. – Christine

Longitudinal sectioning of the coleus leaf, Plectranthus scutellarioides. In the image, you can see the lignified tracheary
elements which were stained with Phloroglucinol-HCL. I would have to say that this image was my favorite. Although it took multiple times for me to get a good view, it was worth it to see the unique spring looking structures in this leaf.
-Bryana Rounds
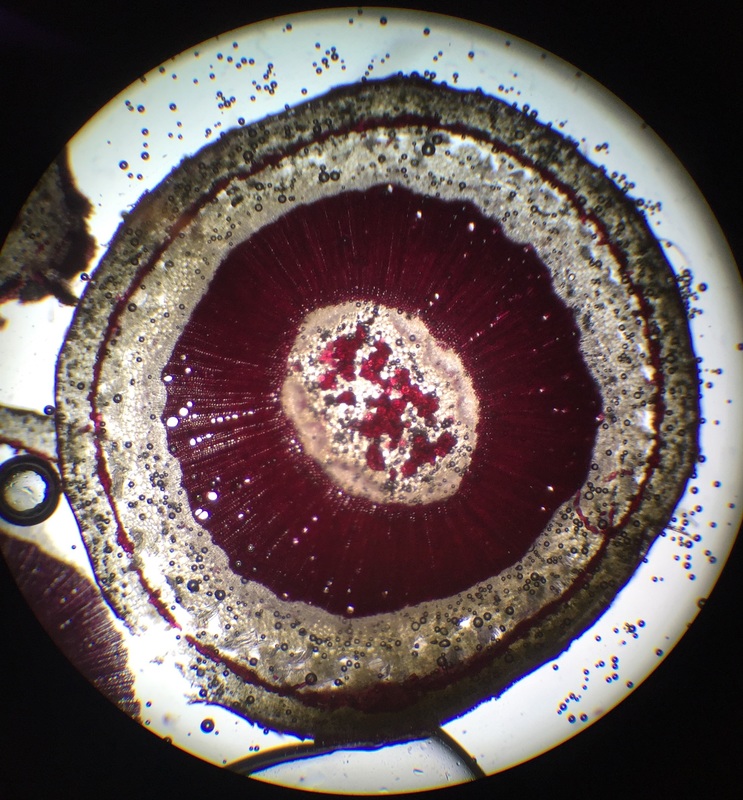
Cross-section of wax plant, Hoya carnosa, stem stained with phloroglucinol- HCl for lignin detection. Lignin is found in the secondary cell wall and turns a red-scarlet red color when stained with phloroglucinol- HCl. From innermost area of the cell - outward, we see brachysclerids in the pith, tracheary elements, and brachysclerids in the cortex. Image taken at 40x total magnification under the compound microscope. This was my favorite specimen to prepare simply because it's beautifully stained. – Christine
GMO Investigations, Part One, Week 5, 2/22/2016
Objectives:
- explain what a genetically modified organism is.
- describe the process of extracting DNA from store-bought food products.
- describe how polymerase chain reaction amplifies target DNA sequences.
- discuss some of the implications for genetic engineering of agricultural crops, both positive and negative, in regards to plant biodiversity, ecosystems, co-evolution of plants and their pests, human health, and economics.
|
|
|
The results of the experiment will be obtained next week when we visualize the products of the PCR using gel electrophoresis.
GMO Investigation II, Week 6, 2/29/16
Objectives:
- explain the process of electrophoresis.
- interpret the results of gel electrophoresis of your PCR samples from last week.
- describe how both positive and negative controls were necessary in order to assess the validity of our results for this investigation.
- discuss some of the implications for genetic engineering of agricultural crops, both positive and negative, in regards to plant biodiversity, ecosystems, co-evolution of plants and their pests, human health, and economics.

The gel ready to be processed using electrophoresis. Electrophoresis works by running a charge through the gel, helping drive the negatively charged DNA sample towards the positive end of the gel. If DNA is present, bands will appear under UV light adter the gel has finished being processed. - Michelle
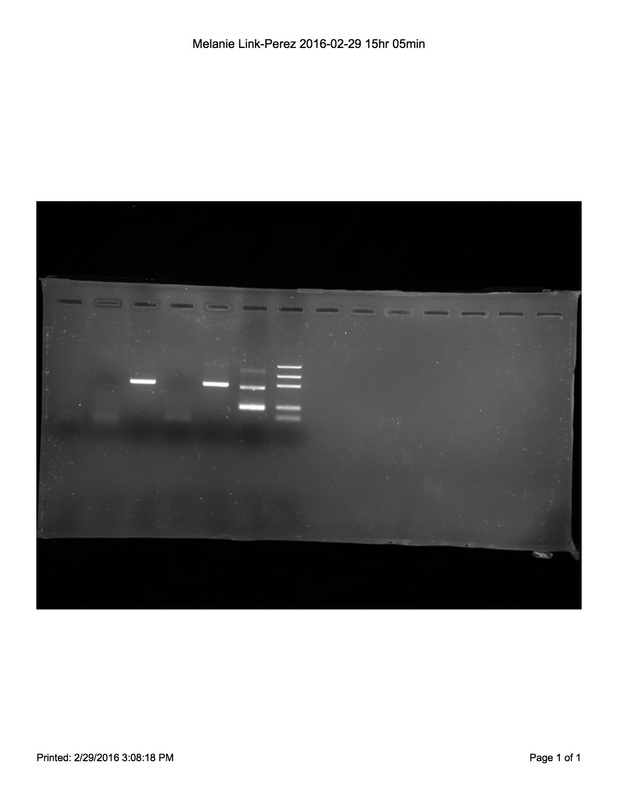
Electrophoresis of PCR products yielded results shown. There is no band in lane 1 (Non-GMO food control with plant primers) or 2 (Non-GMO food control with GMO primers), indicating that we failed to successfully extract DNA from our Non-GMO food control (Certified Non-GMO Oat sample) and our data for that particular sample are inconclusive. Our test food (Smartfood Popcorn) sample revealed a 455 bp long band in lane 3 (test food with plant primers), but no 200 bp band in lane 4 (test food with GMO primers), indicating that our test Smartfood Popcorn sample is Non-GMO. Lane 5 (GMO positive control DNA with plant primers) confirms our results because it too has a 455 bp long band indicating that the sample is from a plant, but does not contain GMOs. Likewise, lane 6 (GMO positive control DNA with GMO primers) contains a 200 bp band that is not shown in lane 4, which further confirms our results. Another group in the lab used the same Smartfood Popcorn as their test food and obtained the same results confirming that Smartfood Popcorn is non-GMO. If we were to repeat the procedure, we would thoroughly grind the Certified Non-GMO oat sample to ensure that DNA may be obtained. - Christine
Bryophytes and Ferns, Week 8, 3/21/16
Objectives:
- recognize representatives of the Phylum Bryophyta and the Phylum Hepaticophyta (non-vascular plants).
- recognize representatives of the seed-free vascular plants- Monilophytes (also known as ferns) at both the microscopic and macroscopic level.
- use the nomenclature, both scientific and common, associated with each non-vascular taxon observed.
- describe the sequence of events that occur during the life cycles of each taxon observed and recognize the gametophyte and sporophyte generations.
- describe the general characteristics of the non-vascular taxa observed and how those characters are used in their classification.

Moss gametophyte and sporophyte. The gametophyte is the leafy region at the bottom of the plant which helps anchor the sporophyte. The sporophyte includes the filaments, known as a seta, connected to the capsule where spores are formed. The capsule is protected by operculum, which falls off to release spores at maturity. The calyptera, a membranous part of the capsule also gives way at maturity, allowing for spore dispersal - Michelle
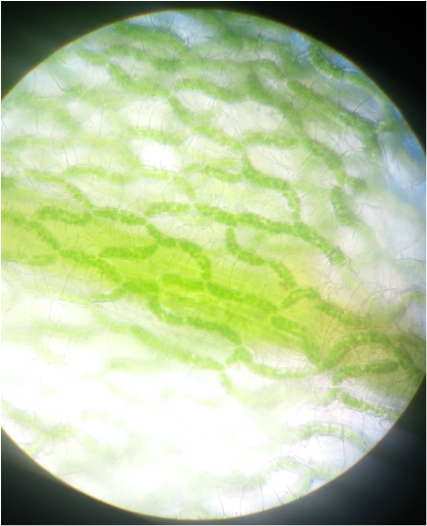
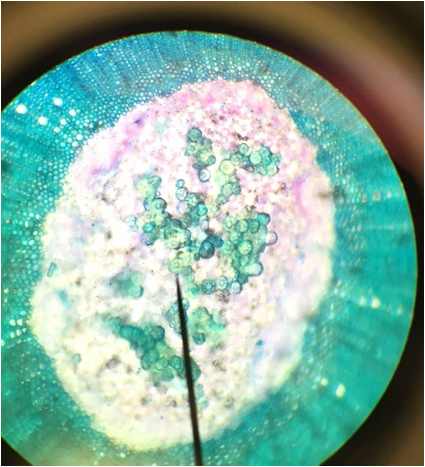
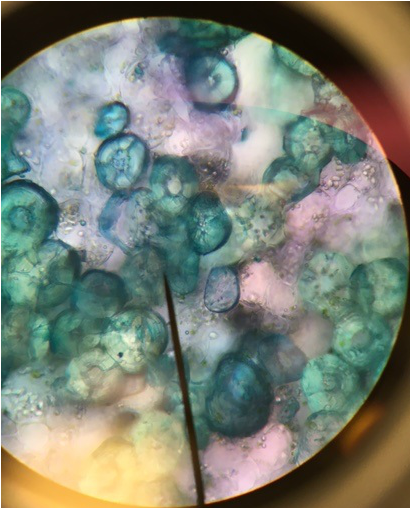
Sphagnum gametophyte. The chlorophyll's cells are indicated by the green chloroplasts found in the branching segments forming a net like structure. This netlike structure surrounds the colorless hyaline cells of the gametophyte. The gametophyte, while appearing leaflike,is undifferentiated tissue unlike true leaves, which are differentiated into dermal, ground and vascular tissue. This is my favorite slide due to the pattern the chlorophylous cells create, as the filaments of cells snake around the hyaline cells- Michelle
|
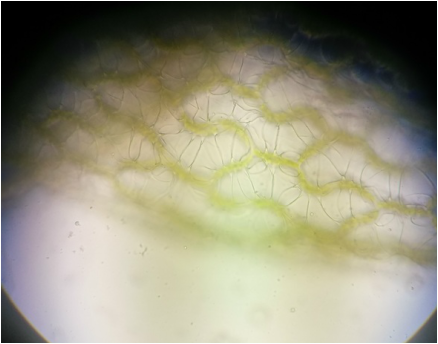
Whole mount of Sphagnum after staining with phloroglucinol to indicate the presence of lignin. After staining, while the color hanged to an orange-green color, no lignin was indicated as it was not stained red. Moss gametophytes lack vascular tissue, and the lack of red pigment from the phloroglucinol HCL indicates this - Michelle
|

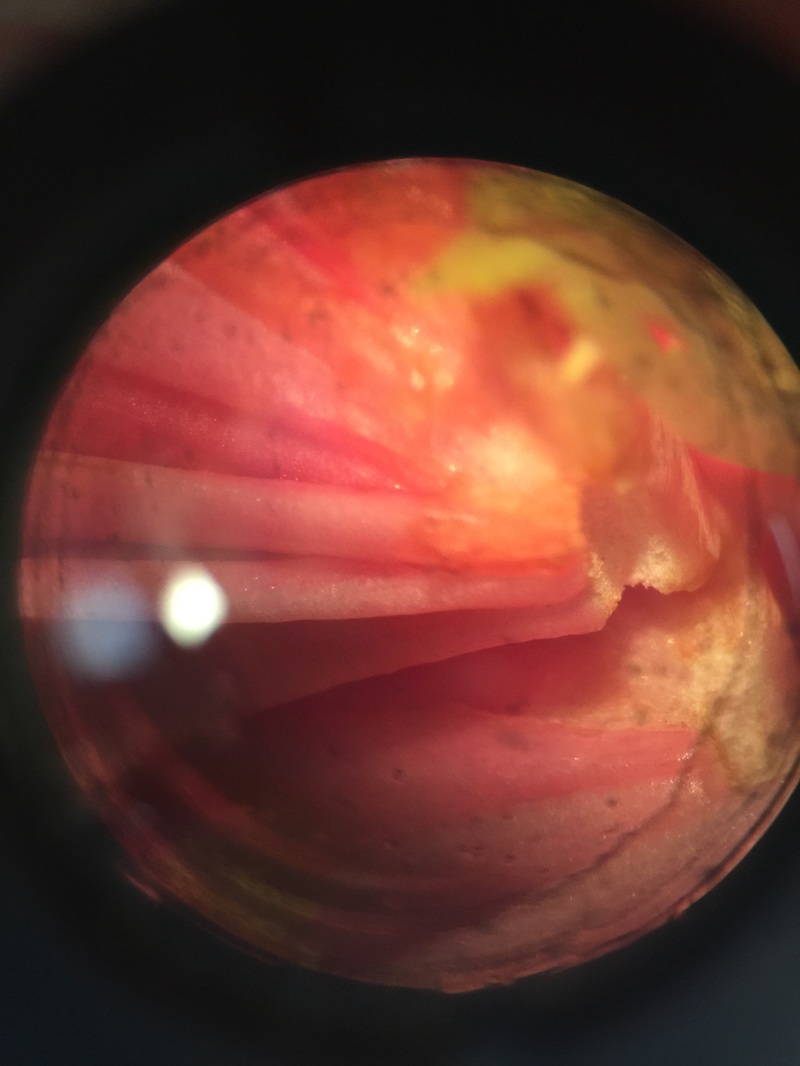
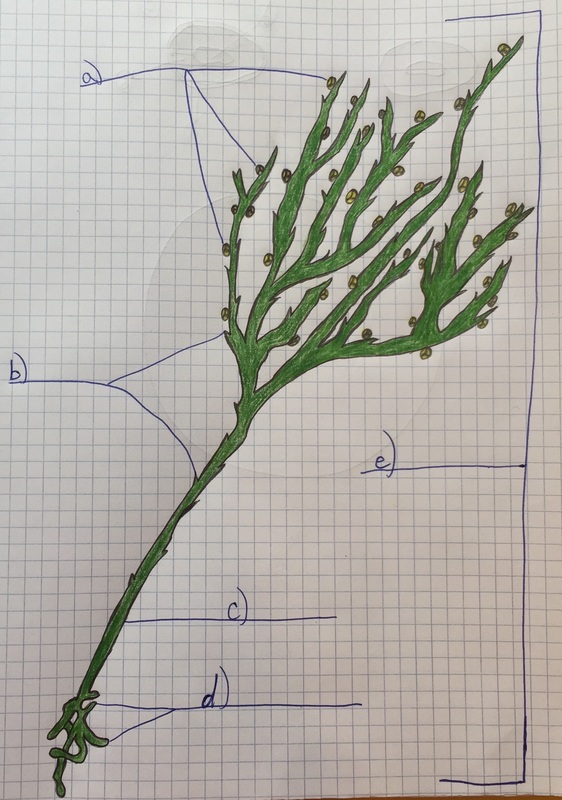
Image of a Nephorrolpsis obliterata, also known as the Australian sword fern which was found in the front of Jenkins Hall on Armstrong's campus. This image was photographed in 20x magnification under the dissecting microscope. The sori on the underside of this fern are in uniform along the backside and don't really compare that much to the sori in figure 17-32.
-Bryana
|

Image of C-fern sperm. This image was photographed in 40x magnification under the compound microscope. There wasn't much movement is my sperm collected, but there was some. The sperm resembles like a motor wheel just spinning around and around. The sperm seemed to only be moving in the same area near the rhizoids. I would have to say this was my favorite image of the day, just because it was interesting watching the release of sperm and the movement of it.
-Bryana
|

Equisetum sp. (horsetail) shown. They are also members of the phylum Monilophyta. Some branching is seen. Internodes and nodes are seen directly under the leaves (brown) of the fertile shoots. These whole structures are mature sporophytes and are diploid. However, no strobili are shown. Sporangia, in which meiosis occurs, would've been found along the margins of the umbrella-like structures called sporangiophores. These sporangiophores are grouped into strobili. - Christine
|

Closeup of Equisetum sp. (horsetail) shown. Branching is better seen. The separation and distinction between the internodes and nodes are more easily seen. The leaves of the fertile shoots are also seen in more detail. Younger leaves are brown. Older leaves appear to be a lighter brown to tannish color. - Christine
|
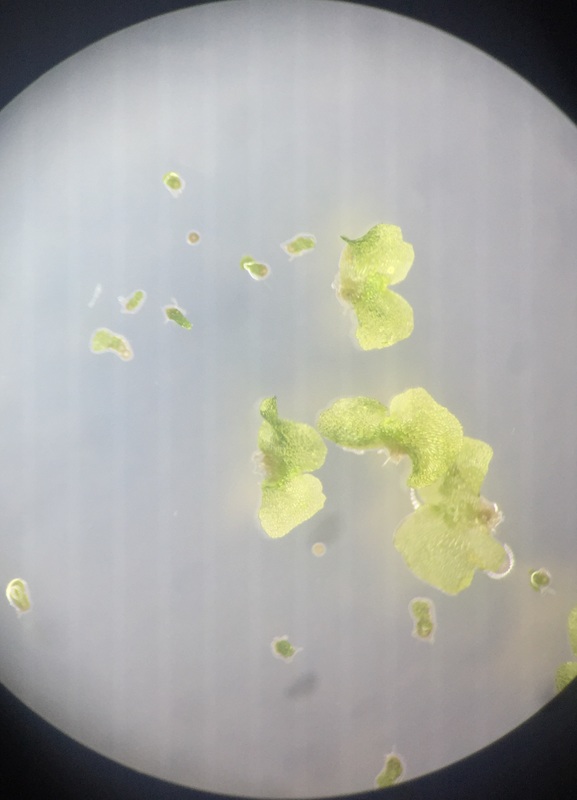
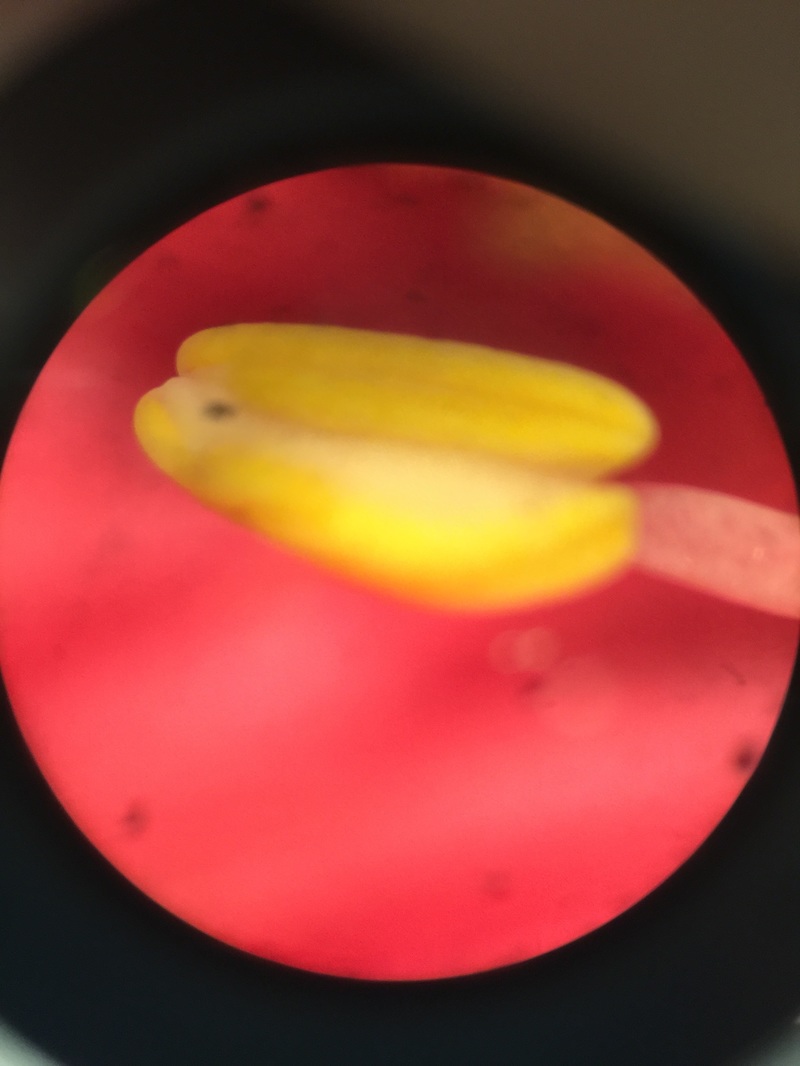
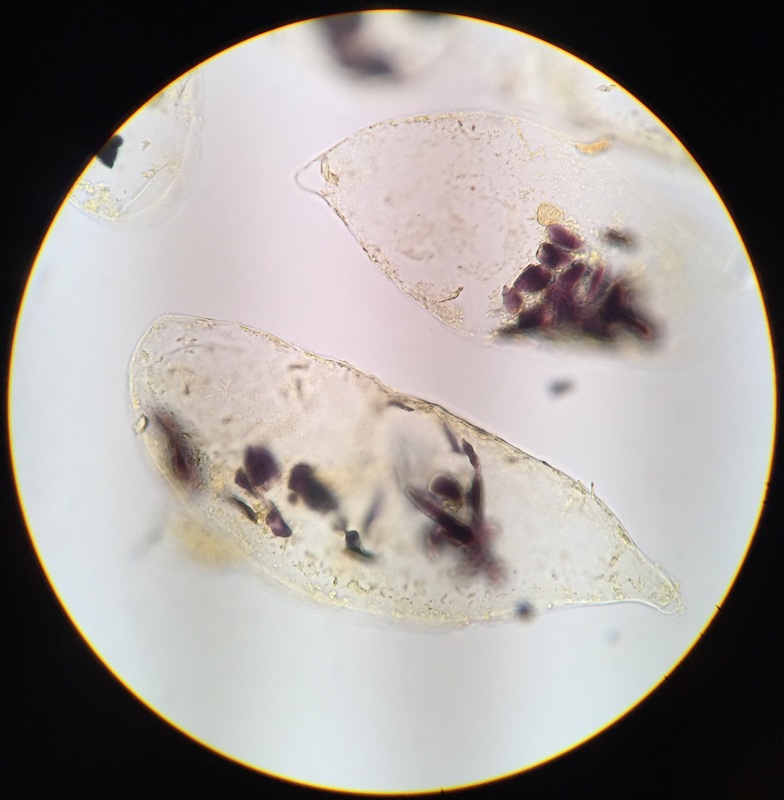
Image shows C-fern gametophytes two weeks after sowing spores on an agar plate viewed under the dissection scope at 30x total magnification. C-ferns are members of the phylum Monilophyta. In this view, we see two small circular brownish colored spores that are ungerminated. The smaller club-shaped gametophytes are male and you can see some of them growing out of the remnants of their spore case. The significantly larger heart-shaped gametophytes are hermaphrodites.
This was my favorite specimen of the day because I sowed these spores and this particular agar plate had everything: a couple of ungerminated spores, several male and hermaphrodite gametophytes, and some male gametophytes growing out of their spore case. - Christine
|
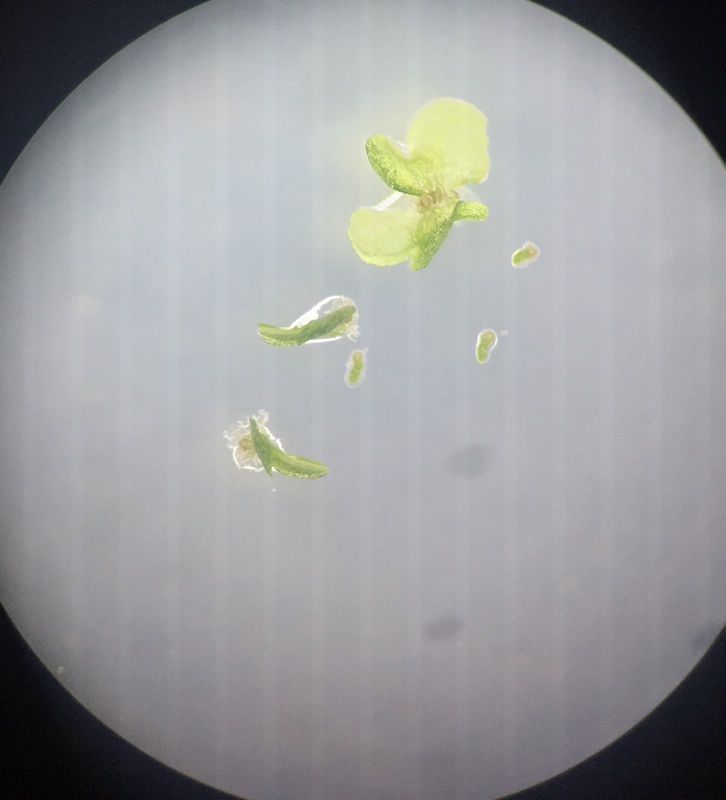
This image also shows C-fern gametophytes two weeks after sowing spores on an agar plate viewed under the dissection scope at 30x total magnification. Here we see three much smaller club-shaped male gametophytes all visibly growing out of the remnants of their spore case and three much larger heart-shaped hermaphrodite gametophytes, two of which are young and appear to be growing sideways. - Christine
|

C-fern sperm shown at 1000x total magnification using oil immersion. It was extremely difficult to try to take a photo of a living sperm cell because they are constantly moving about. The sperm in this image is immobile because it is dead. C-fern sperm are corkscrew shaped and appear to twist (much like a corkscrew) when they move. This sperm sample was obtained by placing a few male gametophytes on a slide with a toothpick. I added a drop of water and a cover slip and watched as the antheridia (mature male sex organ) burst when exposed to the water, releasing their sperm cells. - Christine
Gymnosperms and Angiosperms (Seed Plants), Week 9, 3/28/16
Objectives:
- recognize representatives of the gymnosperms and angiosperms.
- describe the sequence of events that occur during the life cycles of each taxon observed and recognize the gametophyte and sporophyte generations.
- identify the parts of a flower and describe their functions.
- understand the terms related to ovary position, presence and absence of floral organs, and floral symmetry- and be able to use them to describe a specimen.
- prepare diagrams to record important features revealed during a floral dissection.

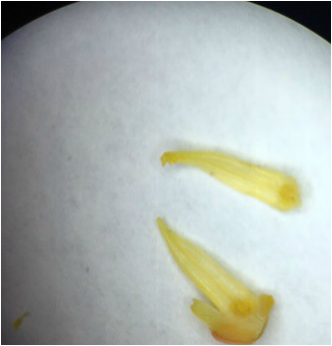
Image of the floral dissection of Dykia 'Burgundy Ice' Burgundy Ice Dykia. This flower is considered zygomorphic or bilateral and it is a complete flower, because it contains sepals, petals, stamens, and carpels. This plant is perfect because it contains both female and male parts. It is also considered a superior ovary, because each whorl is above the ovary. In the image you can see the petals, stamens, sepals, and carpal. This was my most favorite activity of the day, because it was interesting to pull each piece of the flower apart just to get to the inside in order to see the female ovaries.
-Bryana
|

Ginkgo biloba, the only living member of the phylum Ginkgophyta. Also referred to as the maidenhair tree, easily recognized by its fan-shaped leaves with their openly branched, dichotomous pattern of veins, Ginkgo are unique to other gymnosperms because they are deciduous, meaning its leaves turn a beautiful golden color before falling in autumn. - Christine
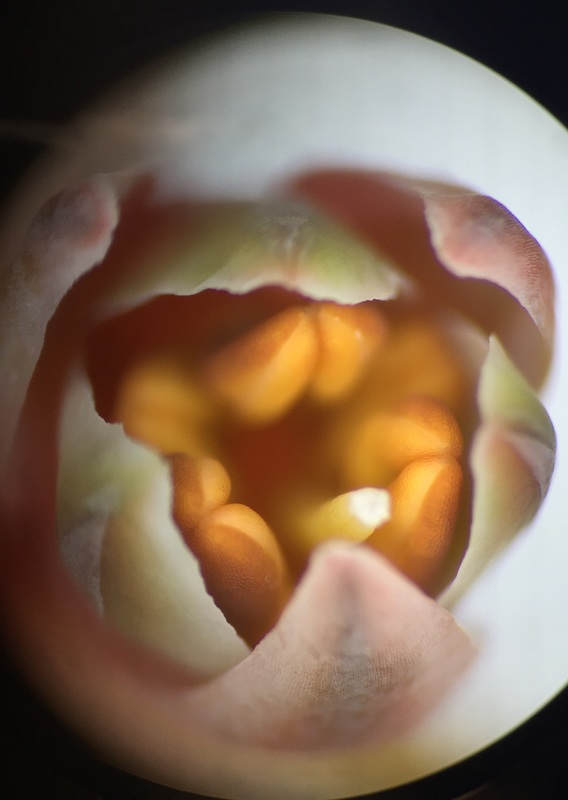
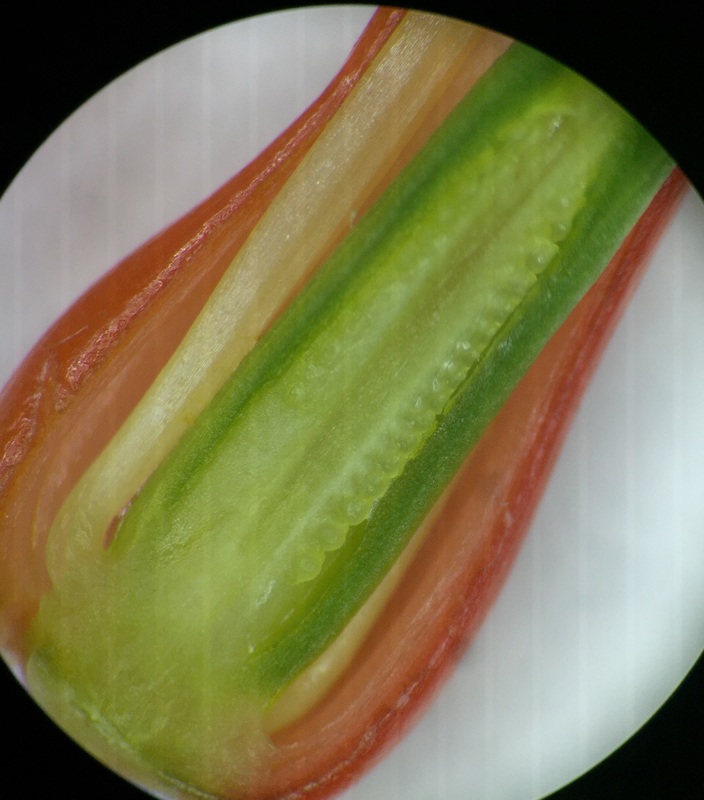
Image shows a top view of an open Aloe maculata bud viewed under 30x total magnification using the dissecting microscope. We see 3 outter sepals protecting the 3 inner petals. We see 6 anthers, the top part of stamens, surrounding the pistil. The stigma is the very top, sticky part of the pistil and the style is the neck region that leads down into the ovary of the flower. The flower is zygomorphic in that it has bilateral symmetry. It is considered incomplete because it does not have 4 whorls. It is perfect because it has both male and female parts and its ovary is inferior. - Christine

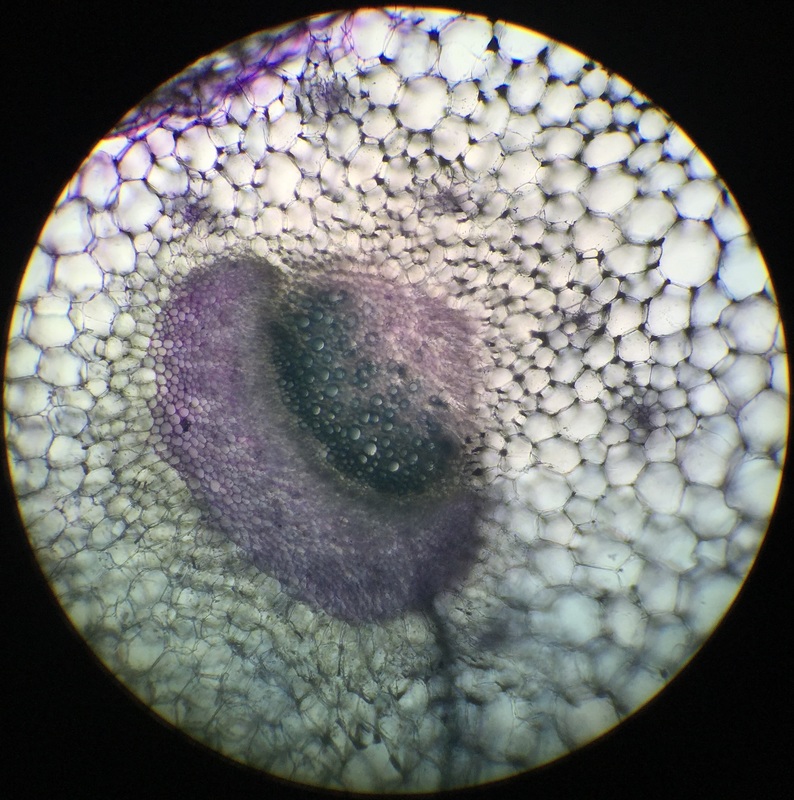
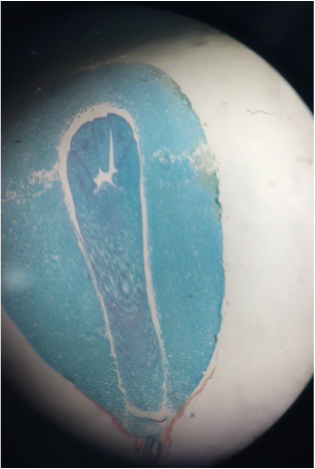
Image shows a longitudinal section of an Aloe maculata carpel viewed under 7.5x total magnification using the dissecting microscope. The very top, sticky part of the pistil is the stigma. The neck region that leads down into the ovary is the style. The whole structure at the bottom is called the ovary, which encases the many ovules we see in this view. The very bottom of the ovary is called the receptacle. This was my favorite specimen of the day, not only because it was fun going out and collecting it, but also because I'm proud of my longitudinal section. - Christine
|
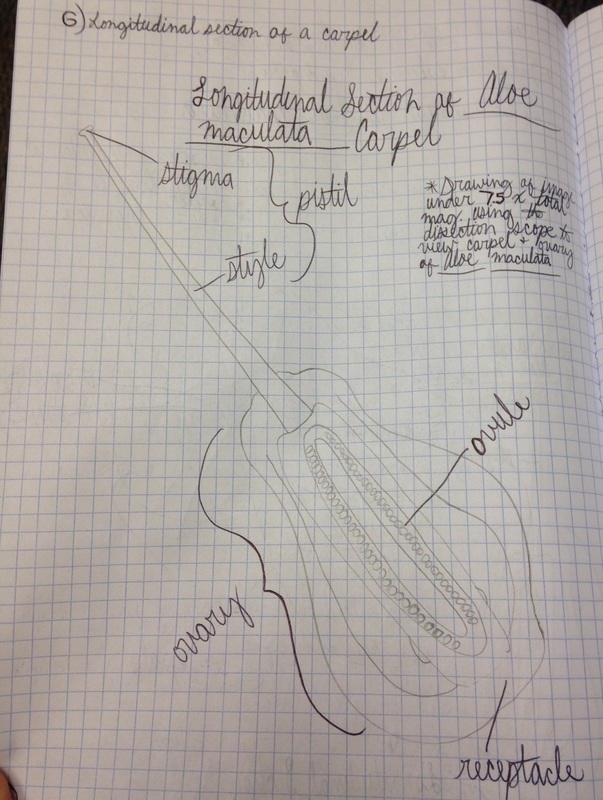
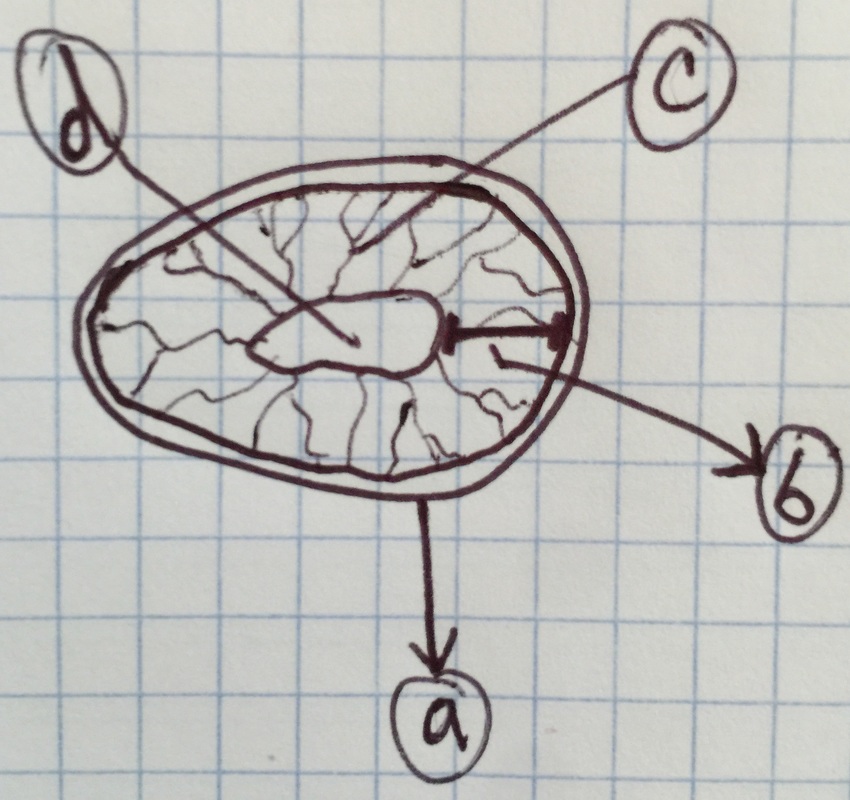
Drawing shows a longitudinal section of an Aloe maculata carpel viewed under 7.5x total magnification using the dissecting microscope. The very top, sticky part of the pistil is the stigma. The neck region that leads down into the ovary is the style. The whole structure at the bottom is called the ovary, which encases the many ovules we see in this view. The very bottom of the ovary is called the receptacle. - Christine
|
Roots and C-ferns Shoot Cuttings and Observations April 11,2016
Today's lab consisted mainly on the focus of roots.
Goal Objectives:
-Identify the external features of plant roots , including root hairs
-Identify the internal anatomy of plant roots
-Distinguish monocot roots from those of dicots, both from external morphology and from internal anatomy
-Describe the principle functions of roots
In today's lab, we worked hands on with identifying the different roots of a monocot (corn) and dicot (pea) seedling. We began a plant root shooting, a culture of cat grass, and transplanted our sporophyte cultures into soil. From today's lab, we have learned the two different types of rooting systems and the internal root anatomy of a monocot and dicot root. Also walked away with learning something, we never knew could happen which is that a plant can grow after cutting its stem.
Goal Objectives:
-Identify the external features of plant roots , including root hairs
-Identify the internal anatomy of plant roots
-Distinguish monocot roots from those of dicots, both from external morphology and from internal anatomy
-Describe the principle functions of roots
In today's lab, we worked hands on with identifying the different roots of a monocot (corn) and dicot (pea) seedling. We began a plant root shooting, a culture of cat grass, and transplanted our sporophyte cultures into soil. From today's lab, we have learned the two different types of rooting systems and the internal root anatomy of a monocot and dicot root. Also walked away with learning something, we never knew could happen which is that a plant can grow after cutting its stem.

Monocot roots of corn plants at different stages of development. In the more mature plant, you can see the shoot-borne or "adventitious" roots growing downward out of the bottom of the shoot. The primary seminal root is growing downward from the corn, but is very difficult to see with the many secondary roots growing out of it. Secondary roots are very important in absorption. The secondary roots of monocots do not run as deep as the secondary roots of dicots. - Christine
|

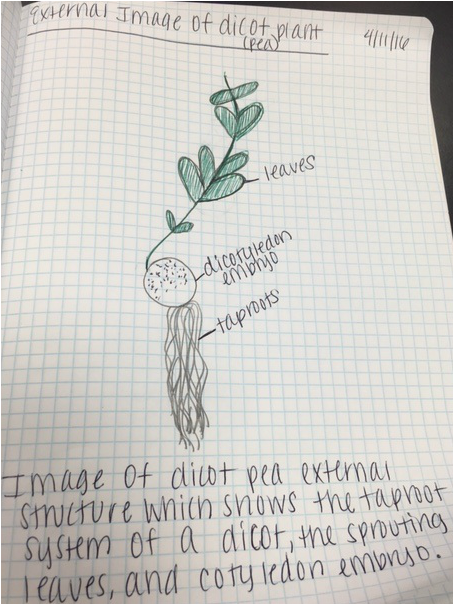
A comparison of the monocot roots of the corn plant and the dicot roots of the pea plant at two different stages of development- early on in development and about 3 weeks into development. In the monocot roots of the corn plant (left), we see many secondary roots growing out of a very difficult to see primary seminal root. We also see the shoot-borne or "adventitious" roots that arise from the stem and give rise to a fibrous root system. In the dicot roots of the pea plant (right), we see the taproot (or primary root), which grows directly downward giving rise to branch/lateral roots (secondary roots), which collectively form the taproot system. - Christine
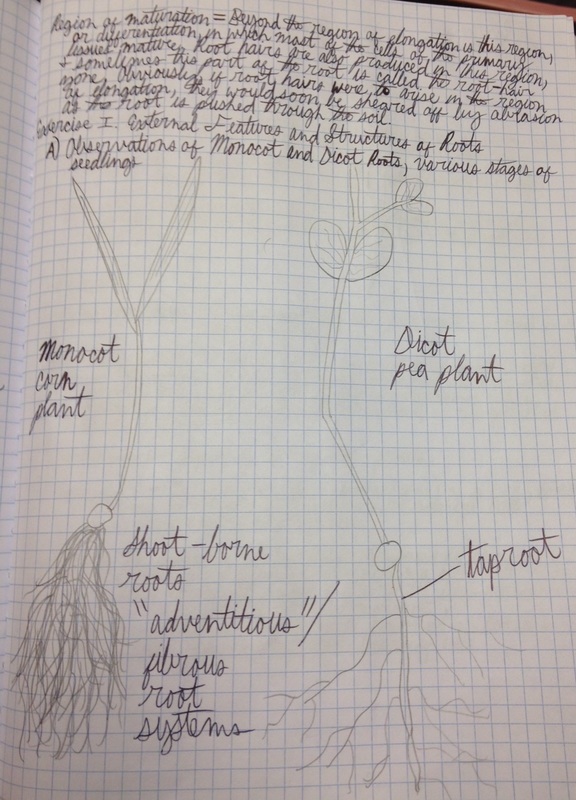
Drawing comparing the monocot roots of the corn plant and the dicot roots of the pea plant at about 3 weeks into development. In the monocot roots of the corn plant (left), we see many secondary roots growing out of a very difficult to see primary seminal root. We also see the shoot-borne or "adventitious" roots that arise from the stem and give rise to a fibrous root system. In the dicot roots of the pea plant (right), we see the taproot (or primary root), which grows directly downward giving rise to branch/lateral roots (secondary roots), which collectively form the taproot system. - Christine

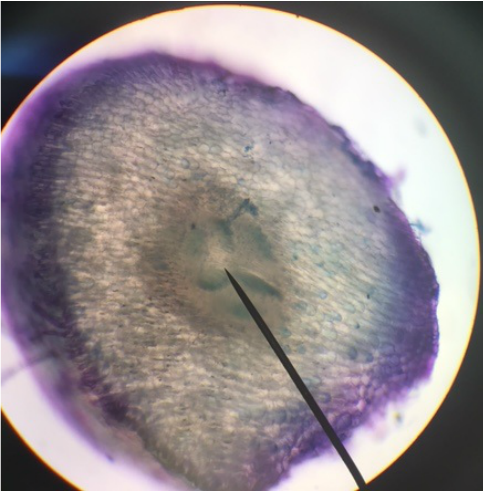
Roots of my C-fern culture at 400x total magnification. The rootcap refers to the yellowish tip of the root. The rootcap is a mass of living parenchyma cells that protects the the apical meristem behind it and helps the root penetrate the soil. As growth occurs, peripheral cells of the rootcap secrete large mounts of mucilage which lubricates the root as it passes through the soil. Eventually, the peripheral cells (border cells) shed from the rootcap and new cells are added to the rootcap. The root hairs are the tubular extensions of the epidermal cells. They greatly increase the absorptive surface of the root. Root hairs are short-lived and confined to the region of maturation. New root hairs are produced just behind the rootcap. - Christine
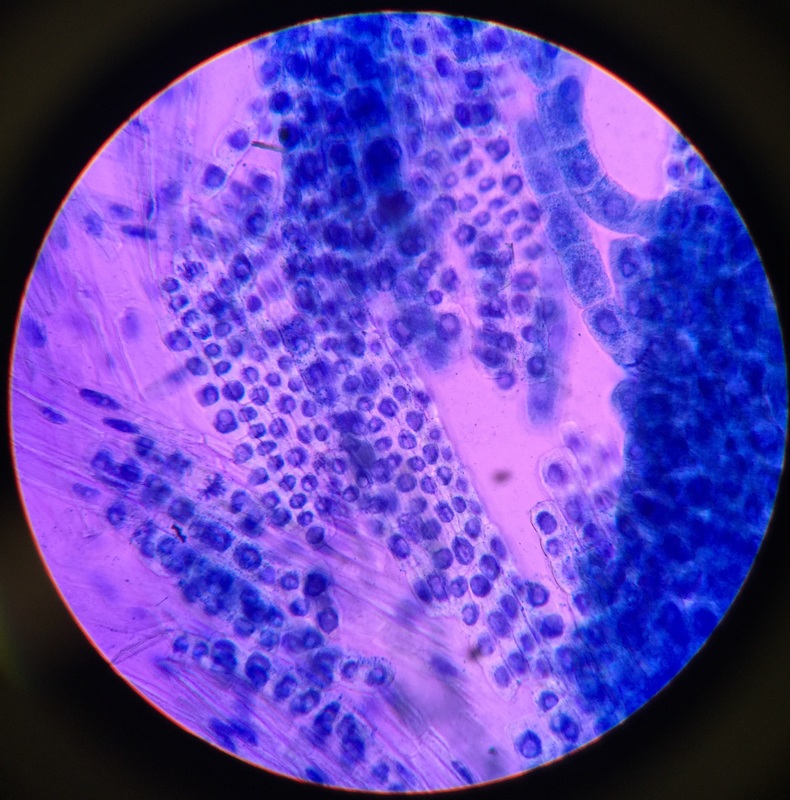
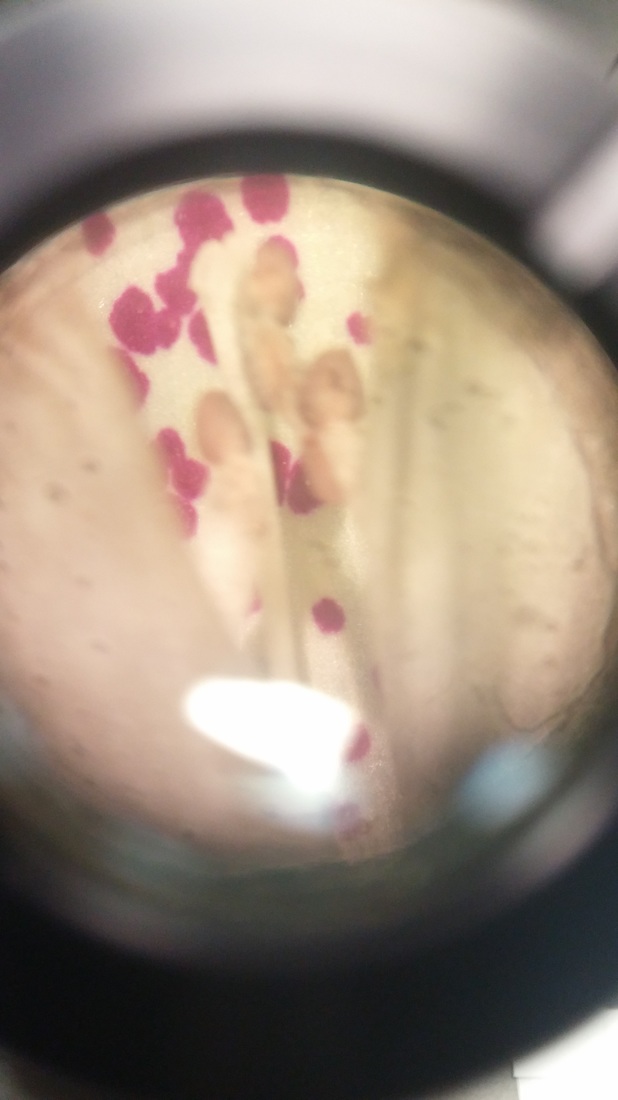
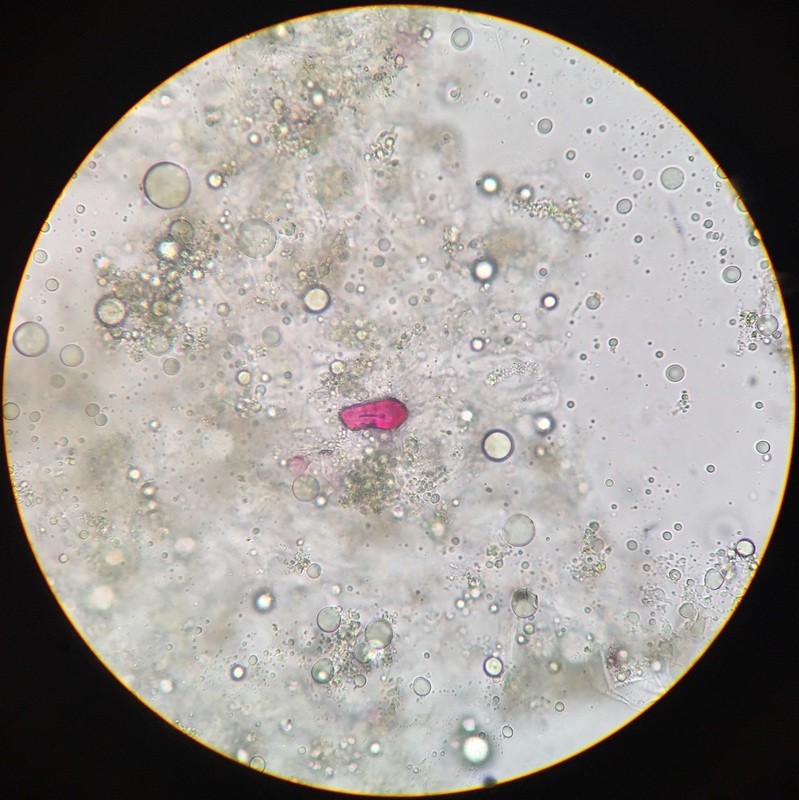
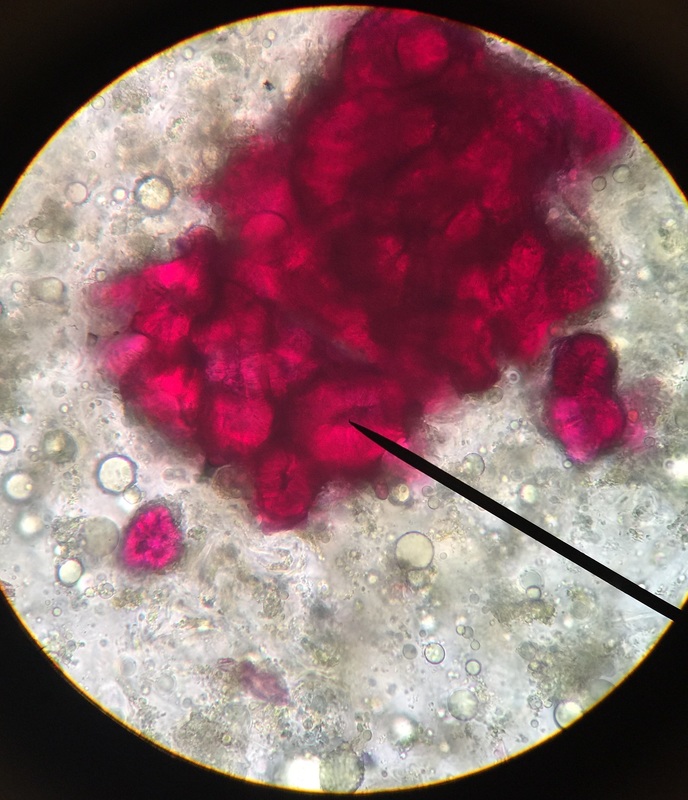
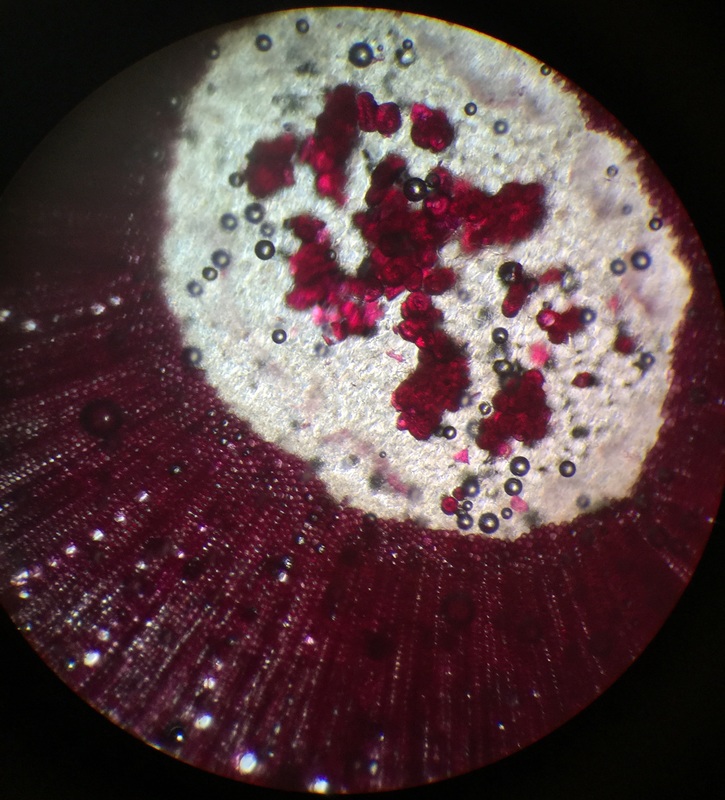
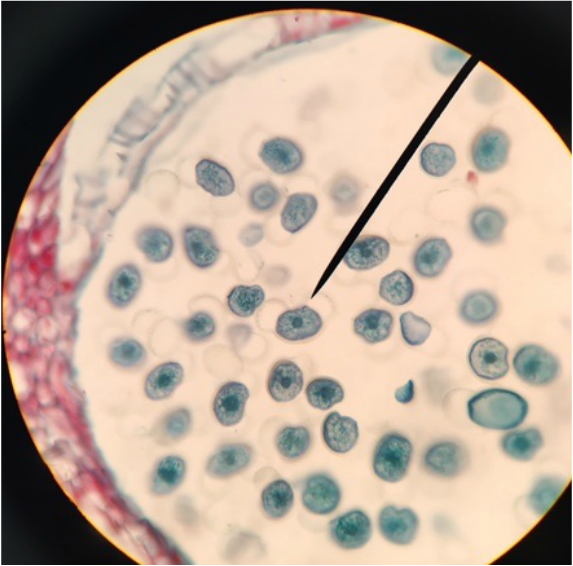
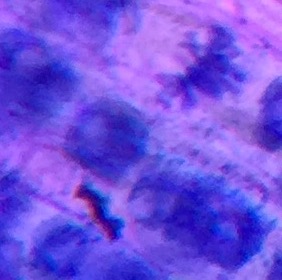
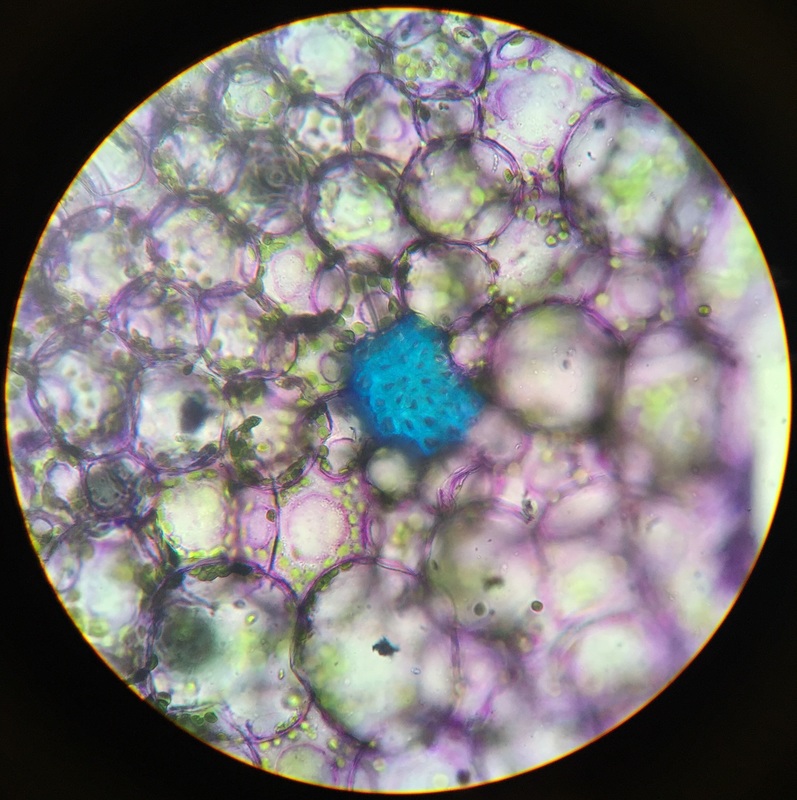
Photo shows mitotic squash of the mitotic cells of Allium cepa (onion) root. Specimen obtained by cutting the apical meristem region of the root, in which growth (elongation) of the root occurs. Speciemen was first put in a solution of Carnoy Fixative I, then in an 18% HCl solution for 4 min., and finally a solution of Carnoy Fixative II for another 4 min. Before staining the tip of the root with TBO to view the mitotic cells, we squashed the specimen onto a slide with a plastic pipette. The two mitotic cells of interest are undergoing metaphase (in quadrant III of the image) in which you begin to see the spindle in the area formerly occupied by the nucleus. During this phase, the chromosomes migrate to the equitorial plane of the spindle. There are two cells undergoing metaphase in quadrant III- one at full metaphase in which the chromosomes lie directly and compactly on the plane of the spindle and the other less compacted on the plane. 400x total magnification - Christine
|
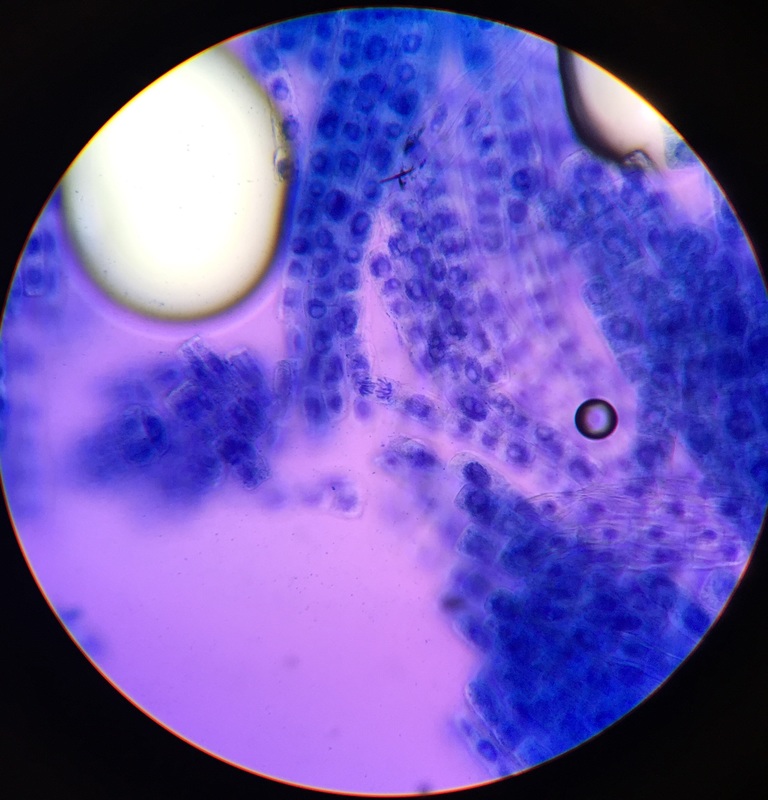
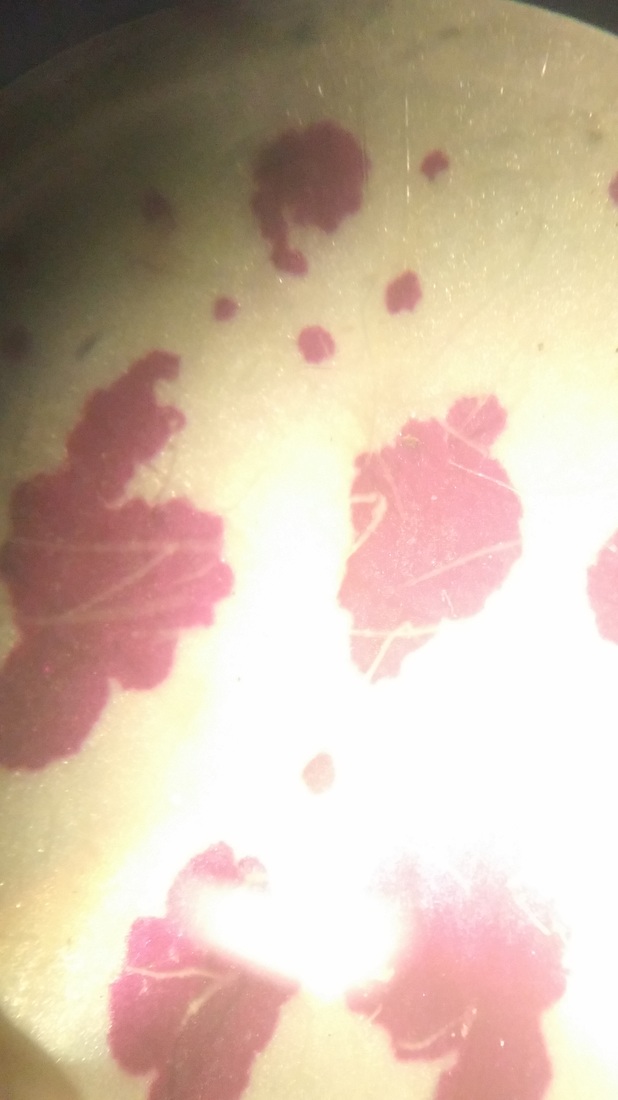
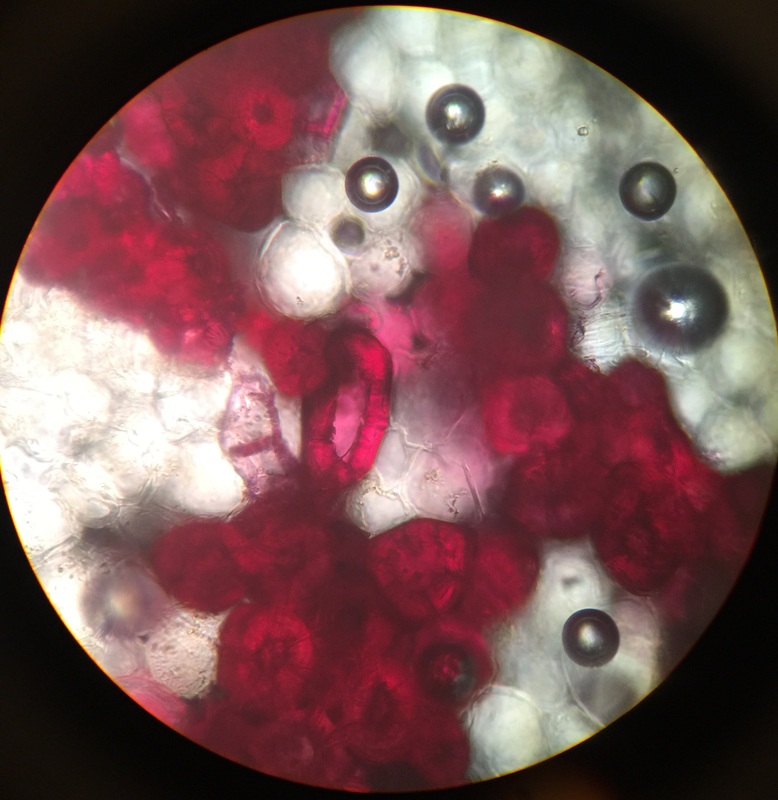
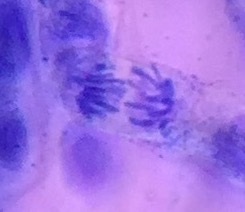
Photo shows mitotic squash of the mitotic cells of Allium cepa (onion) root. Specimen obtained by cutting the apical meristem region of the root, in which growth (elongation) of the root occurs. Speciemen was first put in a solution of Carnoy Fixative I, then in an 18% HCl solution for 4 min., and finally a solution of Carnoy Fixative II for another 4 min. Before staining the tip of the root with TBO to view the mitotic cells, we squashed the specimen onto a slide with a plastic pipette. The mitotic cells of interest are undergoing anaphase (in the very center of the image) in which the centromeres of the sister chromatids separate. The sister chromatids, now daughter chromasomes, then move to opposite poles of the spindle. 400x total magnification. This was my favorite specimen of the day because it was very interesting to capture the mitotic phases of these cells as they were happening in real time. - Christine
|

Image of the C-fern sporophytes after transplating them into the soil. During this process, the sporophytes from our media were removed and put into soil where we will monitor them over the next few weeks in order to track their growth. I would have to say this was my most exciting part of lab, because liked seeeing how much my C-ferns had grown. I cannot wait to see how much they grow over the next few weeks in the soil.
-Bryana