LAB ONE:
Intro to Plant Matter: Dissecting Investigations on Campus
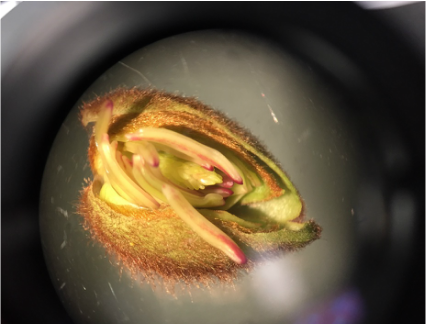
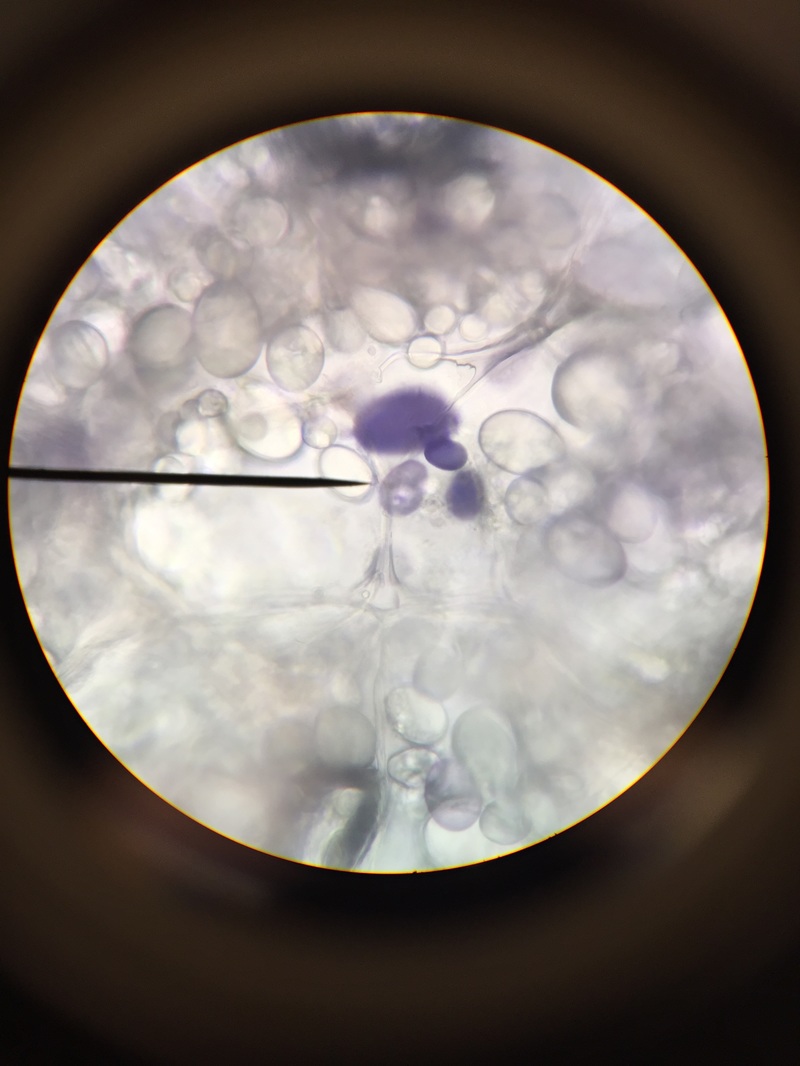
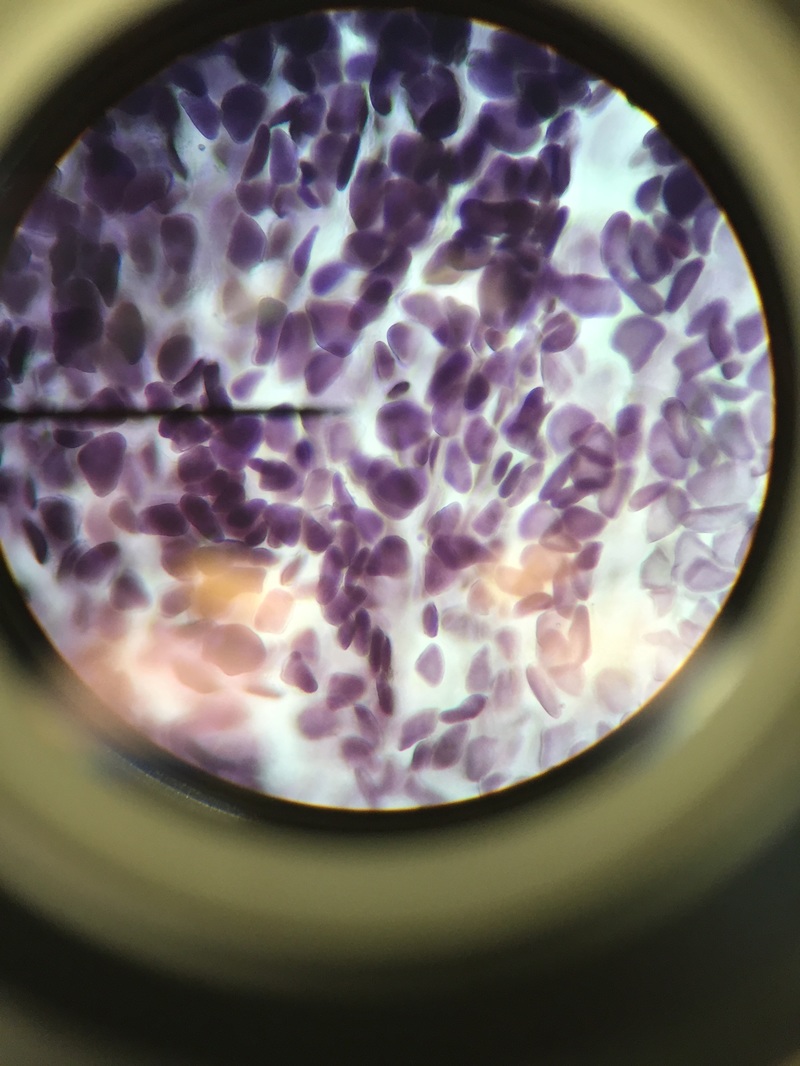
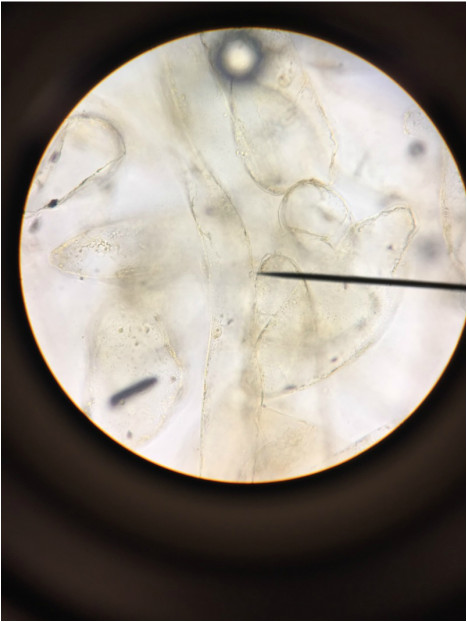
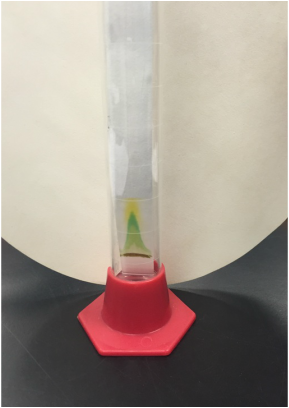
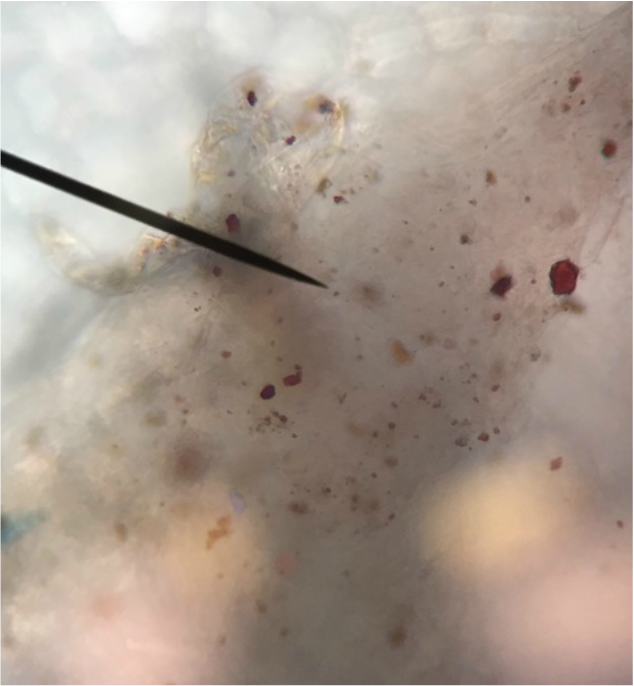
We, Athena and Kelsey, worked as a team to collect five samples of plant matter from the general area of the science centre entrance directly across from the athletics building entrance. We then used the standard ("antique") dissecting microscope to investigate each sample's composition more thoroughly. Each photograph is captioned with the common name and magnification details.
We were surprised to find that, under the dissection microscope, all of the samples, even the stems and scales, were either furry or fibrous in nature.
We then worked together to publish our photos for you to see. Enjoy!
We were surprised to find that, under the dissection microscope, all of the samples, even the stems and scales, were either furry or fibrous in nature.
We then worked together to publish our photos for you to see. Enjoy!
|
|
LAB TWO:
Plant Cells and Scientific Drawings
This week we dove right in to examining parenchyma cells or the principle sites of photosynthesis, assimilation, and storage. We observed many tissue samples from angiosperms (Phylum Anthophyta).
We cut sections from our samples of eight different plants and made slides using a variety of stains and techniques. Sectioning and dying our samples allowed us to highlight the structures inside the tissues. Thus, when we viewed the tissues under our compound microscopes, we were able to observe proplastids, cell walls, cytoplasmic streaming, and much more!
Have a look at what we saw by viewing our photos and scientific drawings, and prepare to be amazed!
"My favourite specimen of the day was seeing the difference between the ripe and un-ripe banana. Seeing the multitude of amyloplast plastids in the un-ripe smear highlighted purple by the Iodine dye versus the stark lack of starches in the ripened smear painted a very clear picture!" -Athena Downes
"My favorite specimen of the day was the Elodea canadensis due to the ease at which one can view the chloroplasts streaming along the edges of the cell wall and cytoplasmic bridges in the cytoplasm. It was also really neat to watch the plant cells begin to shrink after the 5% CaCl solution was added, followed by chloroplast accumulation towards the middle of each cell as the vacuoles loses water due to osmosis, opposite of the cytoplasmic streaming." -Kelsey Hymel
We cut sections from our samples of eight different plants and made slides using a variety of stains and techniques. Sectioning and dying our samples allowed us to highlight the structures inside the tissues. Thus, when we viewed the tissues under our compound microscopes, we were able to observe proplastids, cell walls, cytoplasmic streaming, and much more!
Have a look at what we saw by viewing our photos and scientific drawings, and prepare to be amazed!
"My favourite specimen of the day was seeing the difference between the ripe and un-ripe banana. Seeing the multitude of amyloplast plastids in the un-ripe smear highlighted purple by the Iodine dye versus the stark lack of starches in the ripened smear painted a very clear picture!" -Athena Downes
"My favorite specimen of the day was the Elodea canadensis due to the ease at which one can view the chloroplasts streaming along the edges of the cell wall and cytoplasmic bridges in the cytoplasm. It was also really neat to watch the plant cells begin to shrink after the 5% CaCl solution was added, followed by chloroplast accumulation towards the middle of each cell as the vacuoles loses water due to osmosis, opposite of the cytoplasmic streaming." -Kelsey Hymel
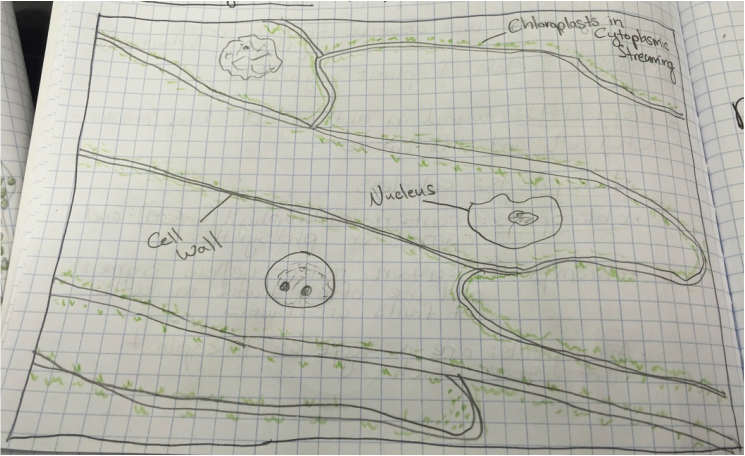
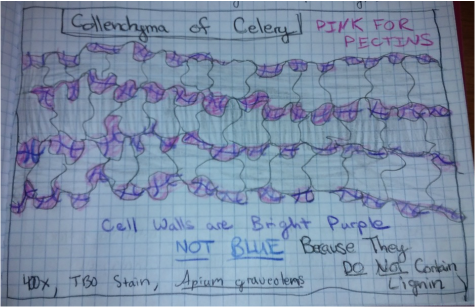
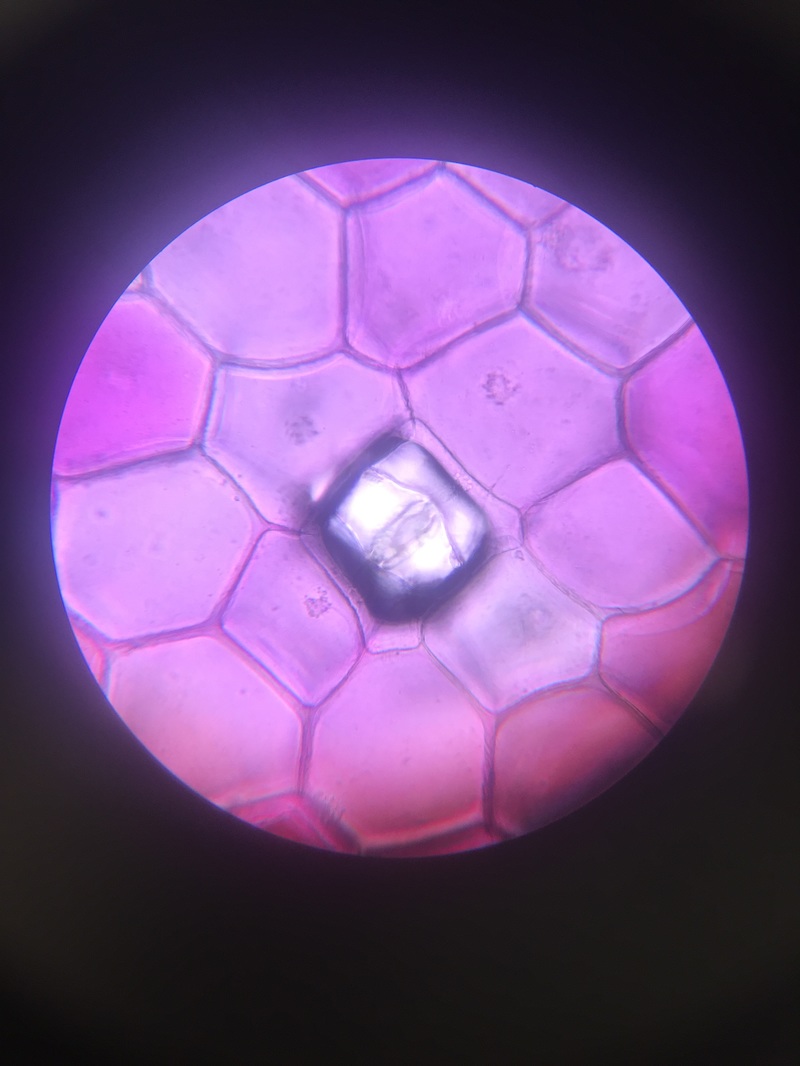
-Sketch by Athena Downes of Identifiable Structures [Cell Wall, Chloroplasts in Cytoplasmic Stream, and Nucleus] as Seen in Epidermis of Allium Cepa (Onion)
-Total Magnification: 400X , Compound Microscope
-Specimen stained with TBO, "Toluidine blue"
-Epidermal Peel Section
-Slide prepared by Athena Downes
-Image by Kelsey Hymel
***NOTE: The TBO stain turns pectic substances a pink-purple colour except for lignin containing walls which turn blue or blue-green. Also the vacuole of each cell takes up appx. 90% of the cell space which makes the nucleus appear to be pushed against the cell wall.***

-Canadian Pondweed Elodea canadensis Cells in 5% CaCl Solution- Image citation: Slide Prepared and Photographed by Brendin Page- ***NOTE: In our procedure we added 5% CaCl salt solution to our Elodea canadensis sample. This caused the cytoplasmic streaming to slow greatly and the cells to move to the middle and skrink. Water cells were moving via osmosis out of the cells. Unfortunately, we got so enthralled in our scope, that we forgot to photo this step! So here is a photo taken by classmate Brendin Page (and posted here with his kind permission) so that you can see what we saw as well!***

-Sketch by Kelsey Hymel of Identifiable Structures [Cell Wall, Cytoplasmic Bridges, Chloroplasts in Cytoplasm, and Inferred Vacuole Space] as Seen in Canadian Pondweed Elodea canadensis Cells
-Total Magnification: 400X , Compound Microscope
-Specimen not stained
-Wholemount Section
-Slide prepared by Kelsey Hymel
-Image by Kelsey Hymel
LAB THREE:
Photosynthesis
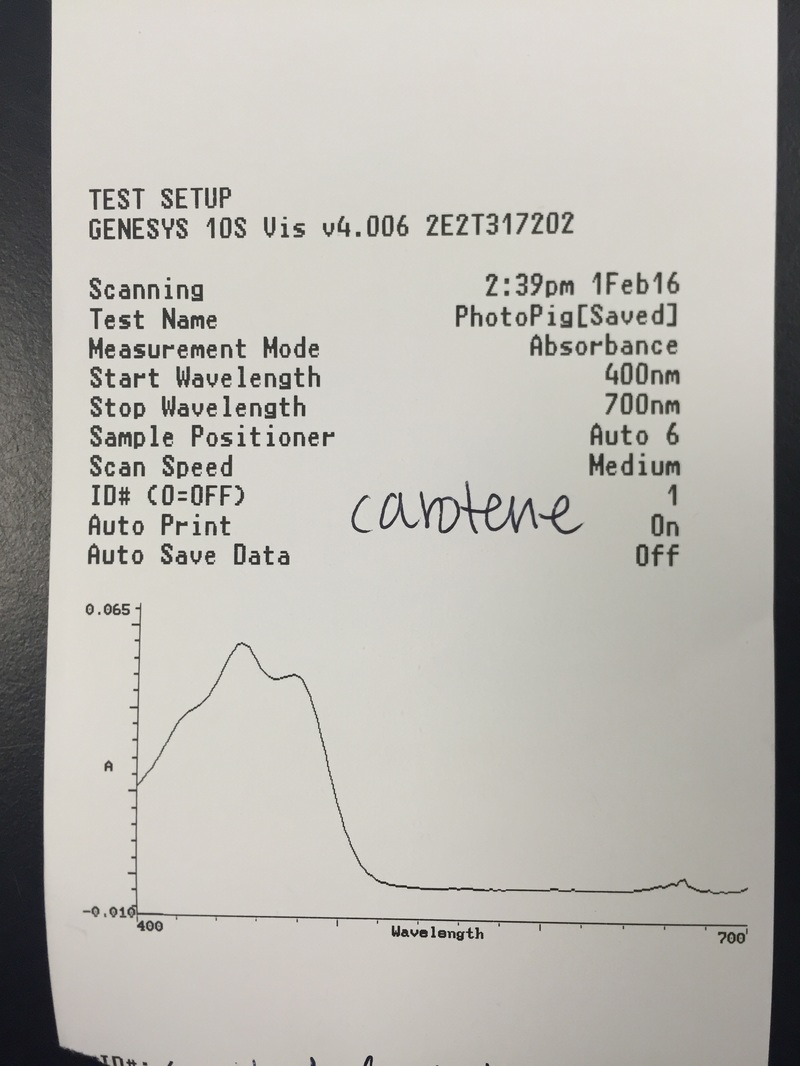
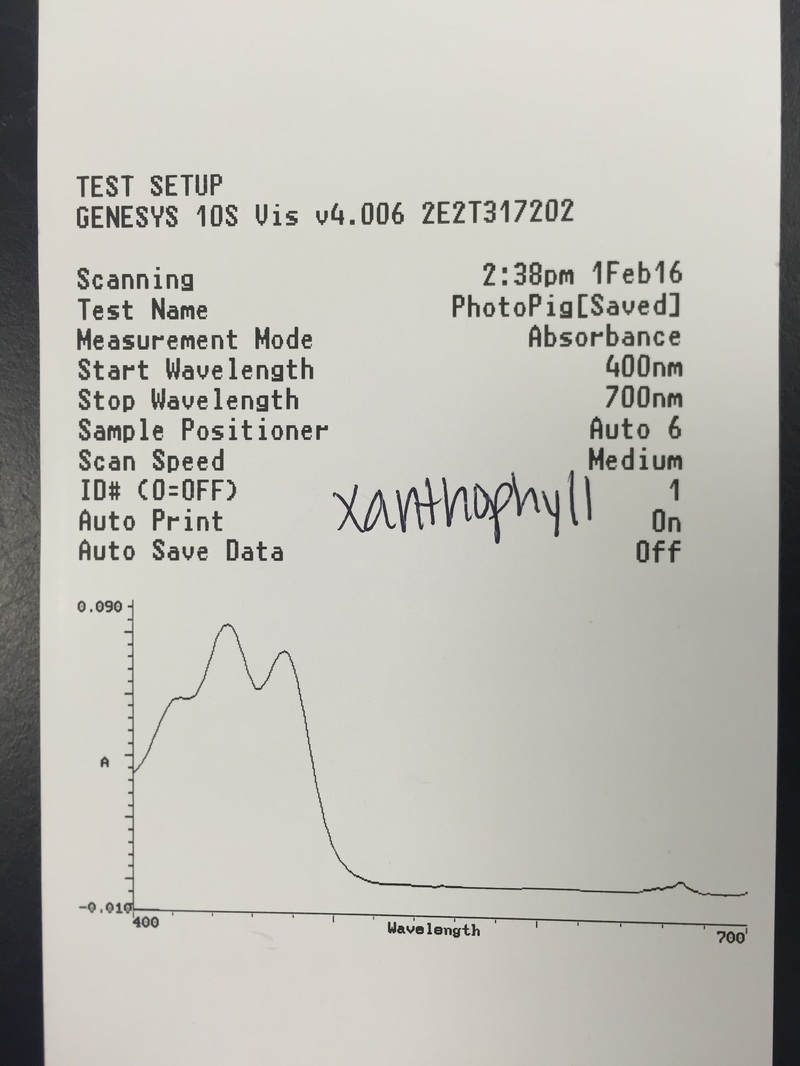
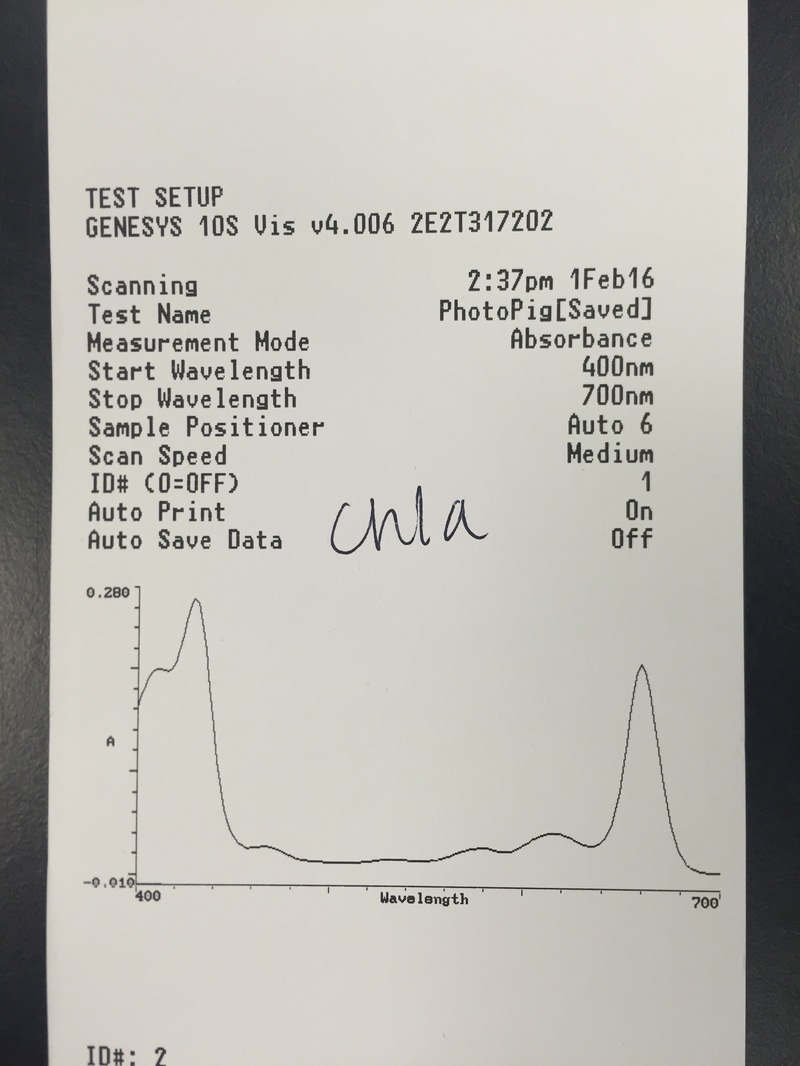
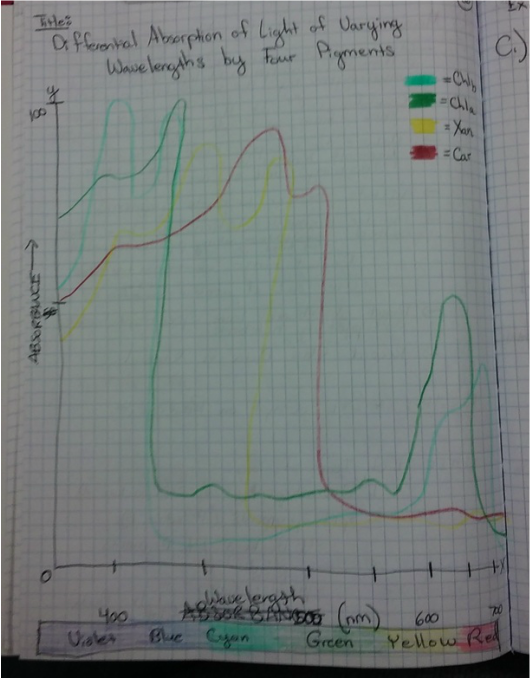
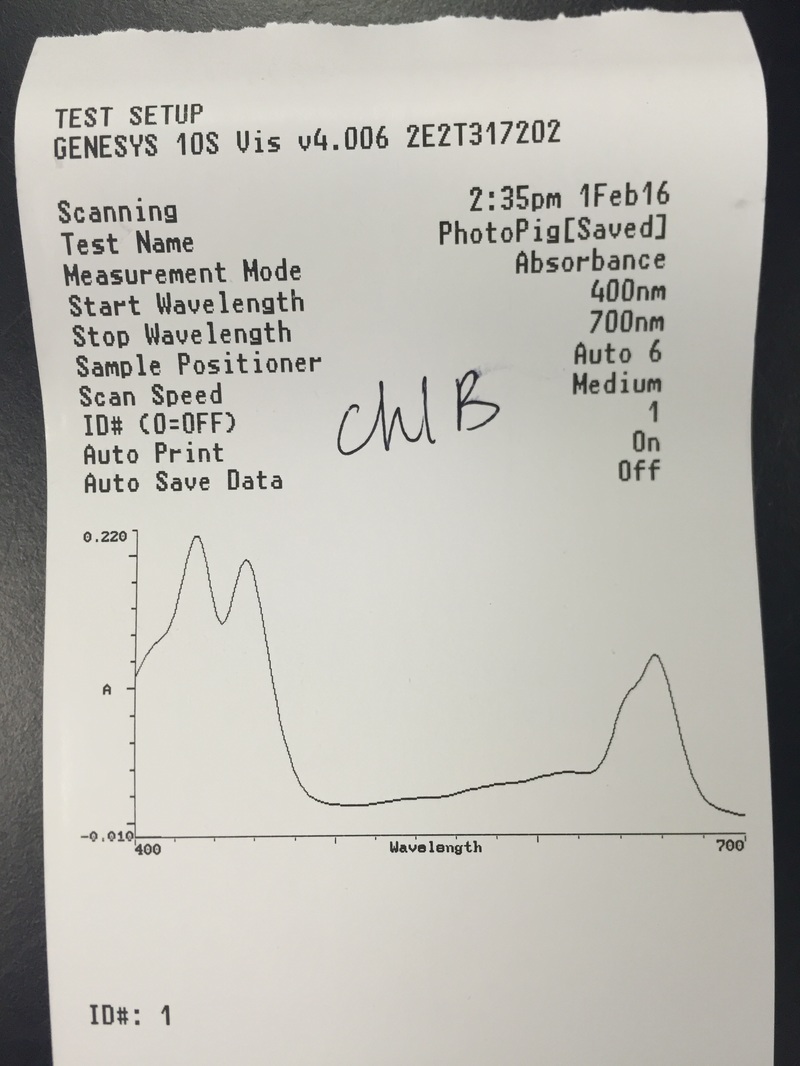
This week we used Armstrong's new spectrometers to measure the differential absorption of light of varying wavelengths by the chloroplast pigments. We extracted and identified the chloroplast pigments of our sample Magnolia grandiflora through chromatography. These processes helped us to visually see the spectrum of absorption that the magnolia plant utilizes by having more than one photosynthetic pigment.
Then, we used Elodea sp. to demonstrate that the "synthesis" reactions are dependent on the products from the "photo" reactions; in other words, in the absence of light- there is no photosynthesis!
Finally, we performed a starch test on leaves from a coleus or Plectranthus scutellariodes to examine the effects of different pigments and photosynthesis.
"My favorite experiment today was extracting and identifying the pigments of the Magnolia grandiflora. It was amazing to see the colors separate out of the crushed leaf cells. Then, using the spectrometers to graph the absorbance of each different color was very clarifying. How fascinating that the entire rainbow spectrum is utilized by the different pigments working in collusion!" -Athena Downes
"Though it was super neat to watch the colors separate out into the different bands on the chromatography paper from the magnolia leaf, my favorite component of this week's lab included the starch test we performed on the leaf of a coleus plant. I watched in awe during Dr. LP's demonstration up front as the pinkish-purple parts of the leaf literally vanished within mere seconds upon being dropped in the boiling water, and I couldn't wait to perform this test on our own once set free. Though the aroma from the boiling ethanol was a bit pungent at times during the second phase of the experiment, it was worth it to watch the green parts of the leaf disappear, turning the leaf translucent before the addition of the iodine. On our particular leaf specimen, it was interesting to see that only ~50% of the leaf was actually photosynthesizing as less than half of the leaf was stained dark by the iodine, noting the presence of starch." -Kelsey Hymel
Then, we used Elodea sp. to demonstrate that the "synthesis" reactions are dependent on the products from the "photo" reactions; in other words, in the absence of light- there is no photosynthesis!
Finally, we performed a starch test on leaves from a coleus or Plectranthus scutellariodes to examine the effects of different pigments and photosynthesis.
"My favorite experiment today was extracting and identifying the pigments of the Magnolia grandiflora. It was amazing to see the colors separate out of the crushed leaf cells. Then, using the spectrometers to graph the absorbance of each different color was very clarifying. How fascinating that the entire rainbow spectrum is utilized by the different pigments working in collusion!" -Athena Downes
"Though it was super neat to watch the colors separate out into the different bands on the chromatography paper from the magnolia leaf, my favorite component of this week's lab included the starch test we performed on the leaf of a coleus plant. I watched in awe during Dr. LP's demonstration up front as the pinkish-purple parts of the leaf literally vanished within mere seconds upon being dropped in the boiling water, and I couldn't wait to perform this test on our own once set free. Though the aroma from the boiling ethanol was a bit pungent at times during the second phase of the experiment, it was worth it to watch the green parts of the leaf disappear, turning the leaf translucent before the addition of the iodine. On our particular leaf specimen, it was interesting to see that only ~50% of the leaf was actually photosynthesizing as less than half of the leaf was stained dark by the iodine, noting the presence of starch." -Kelsey Hymel
Experiment #1 - Plant Pigments
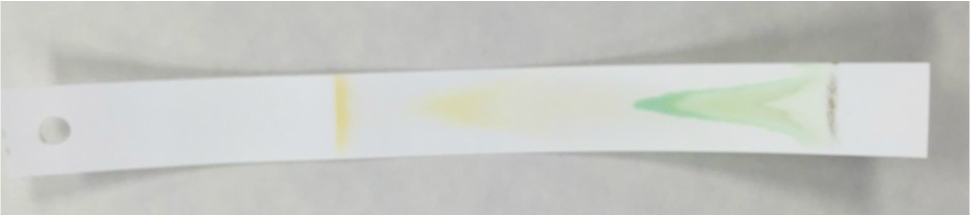
- The Chromotagraphy Paper Showing the Four Different Pigments Contained in the Magnolia grandiflora Leaf Cells - From Left, Carotene [Dark Orange Line], Xanthophylls [Yellow Curve], Chlorophyll a [Dark Blue/Green Curve], and Chlorophyll b [Lighter Green/Lime Curve] *NOTE: The once dark green line of crushed leaf cells now appears brown in color.* - Photograph by Kelsey Hymel
Experiment #2 - CO2 Consumption in Photosynthesis
Experiment #3 - Test for Starch Differences in Pigments
LAB FOUR:
Simple and Complex Tissues
This week we used our burgeoning section making skills to compare and contrast parenchyma, collenchyma, and sclerenchyma cells of the ground tissue system. Also we observed and identified the three tissue systems of the plant body as well as the water-conducting cells of the vascular tissue system while thinking about the importance of their structures to their functions. Finally, we described the characteristic of the epidermis.
*NOTE: All scientific facts about the different cells types and functions that are referenced on the images are from class materials.*
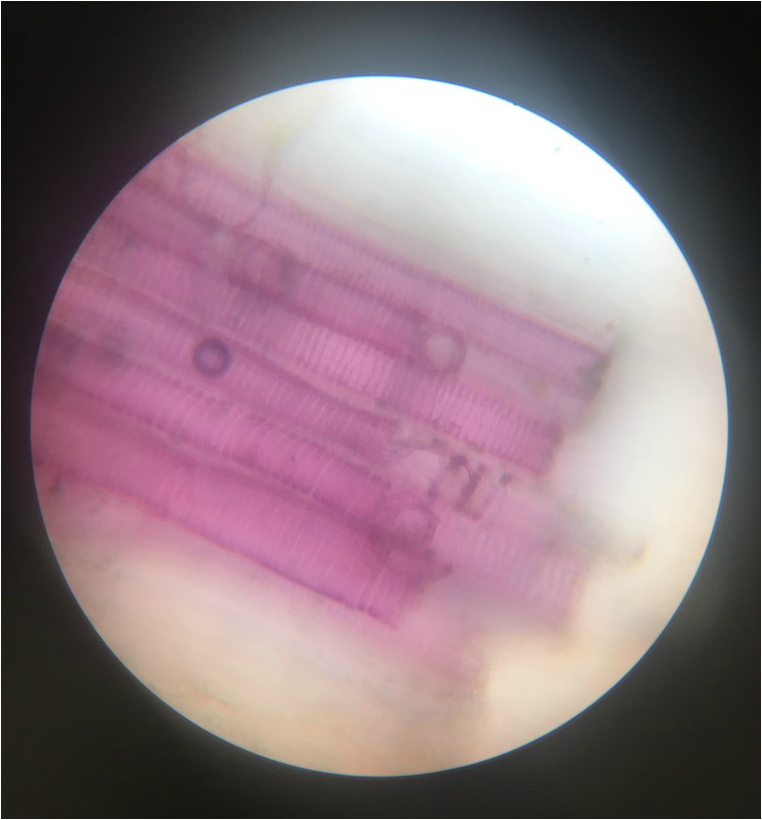
"I have to say this lab was the most fascinating photographically thus far! I am continually amazed by the intricate and fabulous microscopic world of plant cells, and I am so pleased to be able to share our photos with you. My favourite specimen of this lab was the Apium graveolens (celery petiole) because of the variety of cells, colours, and functions that could be seen in the one tiny section!" -Athena Downes
"This has been my favorite lab to date! I love preparing slides and staining slides in general, but to be rewarded by such awesome looking specimens was a special bonus. I would never be able to guess in a million years that these all these photographs came from plant tissues and cells. They are all fascinating, but I do have to say my favorite specimen was the sclerenchyma fibers of the snake plant, Sansevieria trifasciata, the TBO stained the fibers such a bright, electric blue that looked more like a spider's web than a cross-section of fibers in a common plant." -Kelsey Hymel
*NOTE: All scientific facts about the different cells types and functions that are referenced on the images are from class materials.*
"I have to say this lab was the most fascinating photographically thus far! I am continually amazed by the intricate and fabulous microscopic world of plant cells, and I am so pleased to be able to share our photos with you. My favourite specimen of this lab was the Apium graveolens (celery petiole) because of the variety of cells, colours, and functions that could be seen in the one tiny section!" -Athena Downes
"This has been my favorite lab to date! I love preparing slides and staining slides in general, but to be rewarded by such awesome looking specimens was a special bonus. I would never be able to guess in a million years that these all these photographs came from plant tissues and cells. They are all fascinating, but I do have to say my favorite specimen was the sclerenchyma fibers of the snake plant, Sansevieria trifasciata, the TBO stained the fibers such a bright, electric blue that looked more like a spider's web than a cross-section of fibers in a common plant." -Kelsey Hymel
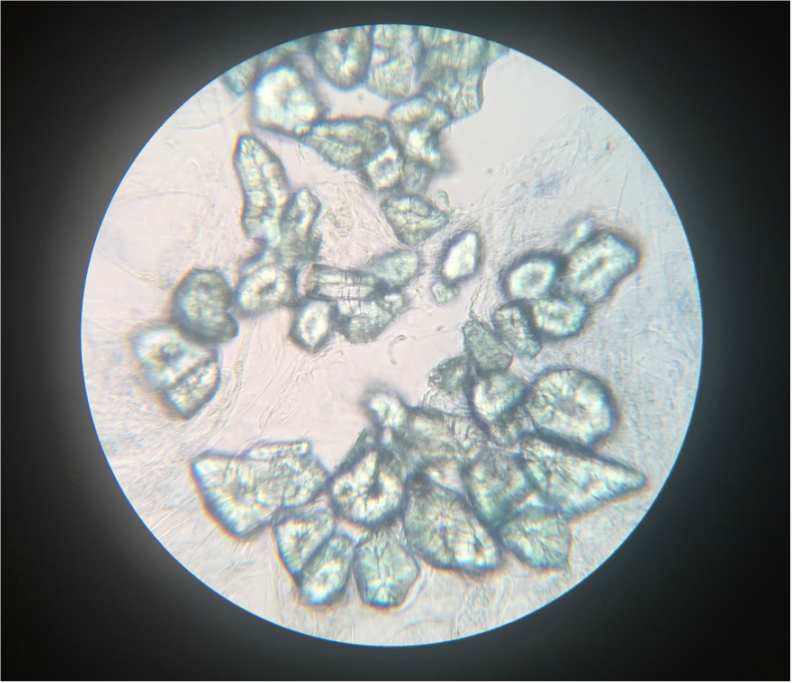
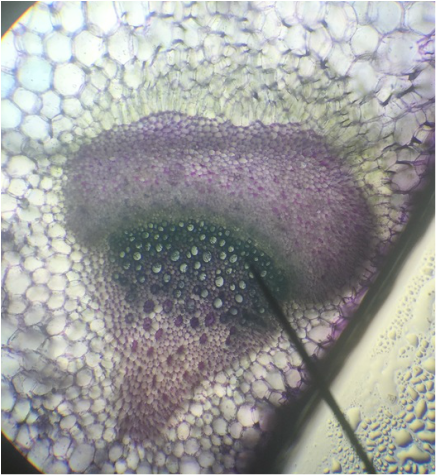
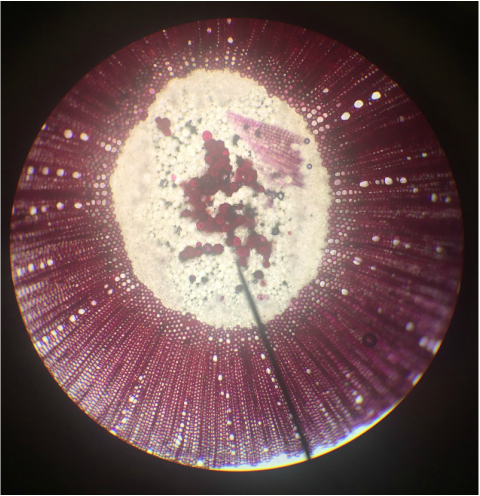
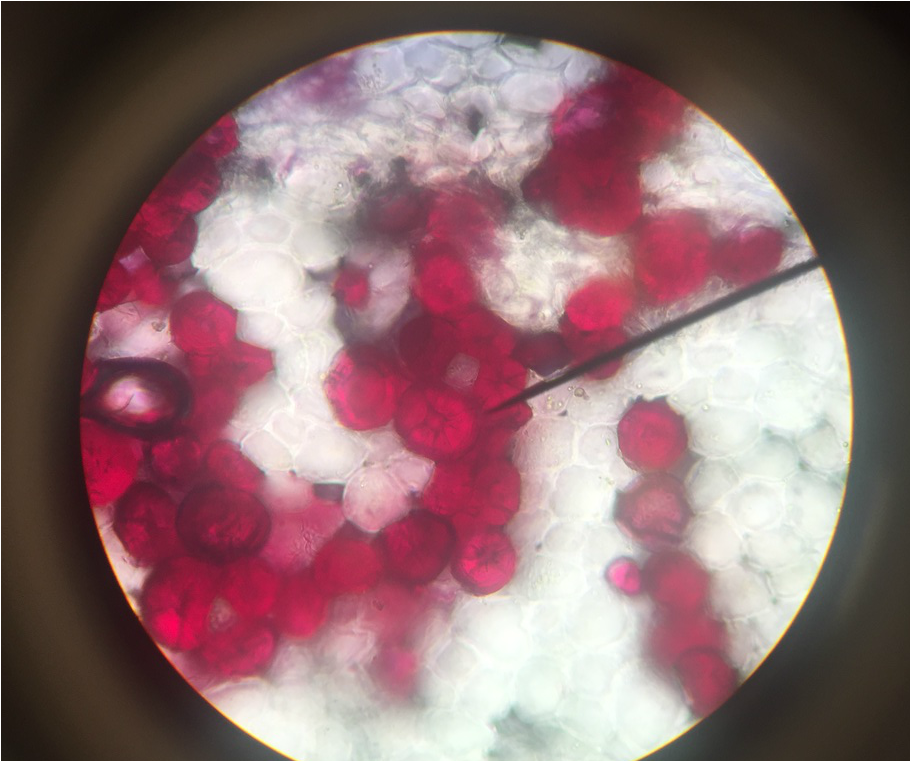
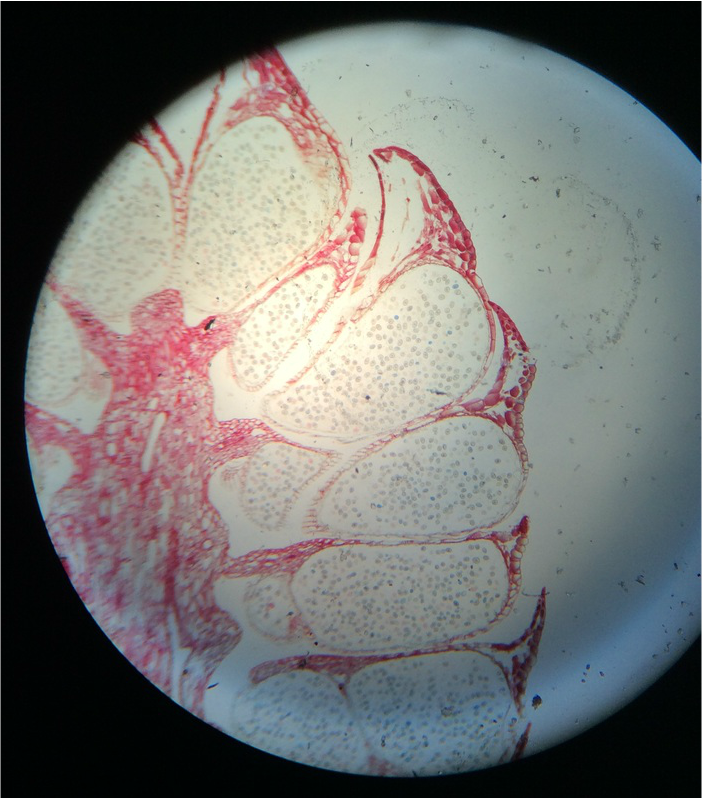
- Structural Support Cells Called Brachysclereids or "Stone Cells" in Sclerenchyma Cells of Pyrus communis , Pear Fruit, Showing Lignified Cell Walls and Pit Canals - Stained with TBO - Sectioned Sample - Total Magnification 400x - Slide prepared by Kelsey Hymel - Photo Credit: Kelsey Hymel ***NOTE: Stone cells are the cells which give pears their gritty texture.***
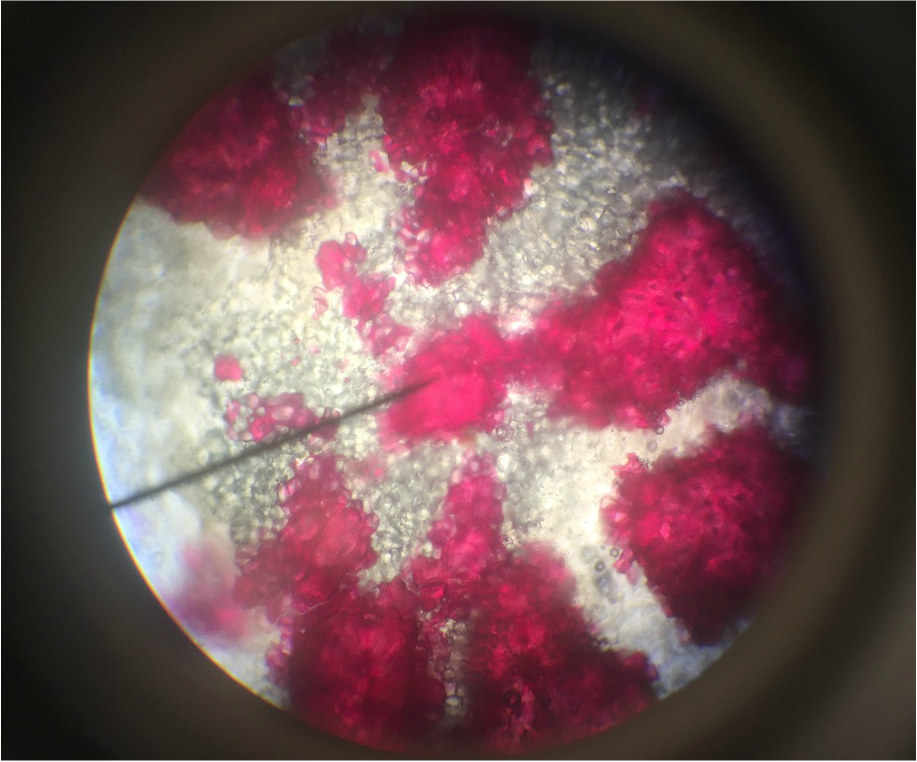
- Sclerenchyma Cells of Persea americans , Avocado Fruit, Showing Lignified Cell Walls and Brachyscleriod Stone Cells with Pit Canals - Stained with phloroglucinola-HCL - Sectioned Sample - Total Magnification 400x - Slide prepared by Athena Downes - Photo Credit: Athena Downes ***NOTE: Lignified cell walls appear various shades of red depending on type and amount of lignin; due to the intensity of this colour we can infer presence of a significant amount of lignin in the secondary cell walls of the brachysclereids.***
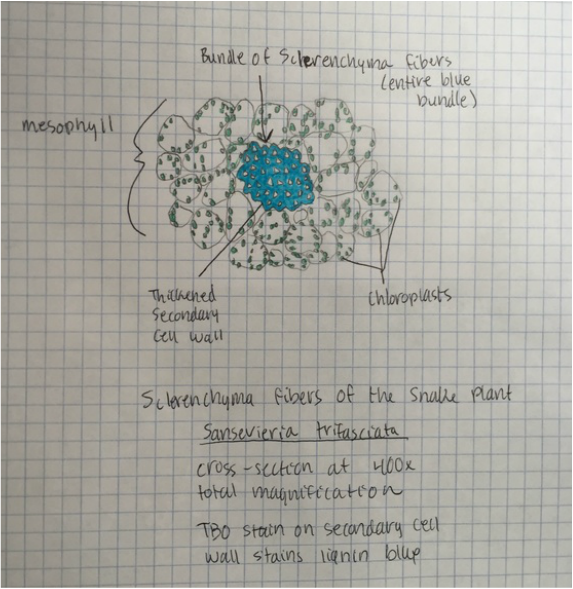
- Representation of Sclerenchyma Fibers of Snake Plant, Sansevieria trifasciata , Showing Bundle of Fibers in the Mesophyll , Thickened Secondary Cell Walls (Bright Blue), and the Green Chloroplasts- Stained with TBO - Cross-Sectioned Sample - Total Magnification 400x - Drawing by Kelsey Hymel - Photo Credit: Kelsey Hymel ***NOTE: The secondary cell wall stains bright blue or blue green because of the presence of thickened layers of ligin.***

- Sclerenchyma Fibers of Snake Plant, Sansevieria trifasciata , Showing Bundle of Fibers in the Mesophyll , Thickened Secondary Cell Walls (Bright Blue), and the Green Chloroplasts- Stained with TBO - Cross-Sectioned Sample - Total Magnification 400x - Slide prepared by Kelsey Hymel - Photo Credit: Kelsey Hymel ***NOTE: The secondary cell wall stains bright blue or blue green because of the presence of thickened layers of ligin.***
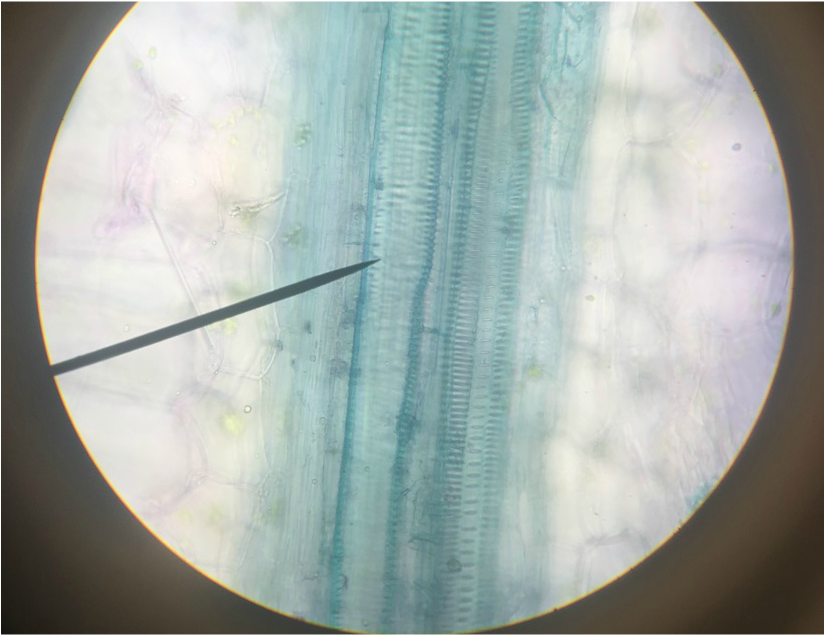
- Sclerenchyma Fibers of Snake Plant, Sansevieria trifasciata , Showing Narrow, Elongated Shape of the Supportive Fibres in the Xylem- Stained with TBO -Longitudinal Section - Total Magnification 400x - Slide prepared by Athena Downes - Photo Credit: Athena Downes***NOTE: The secondary cell wall stains bright blue of blue green because of the presence of thickened layers of login. Annular, spiral, and scalariform secondary cell wall structures can be seen in this image.***
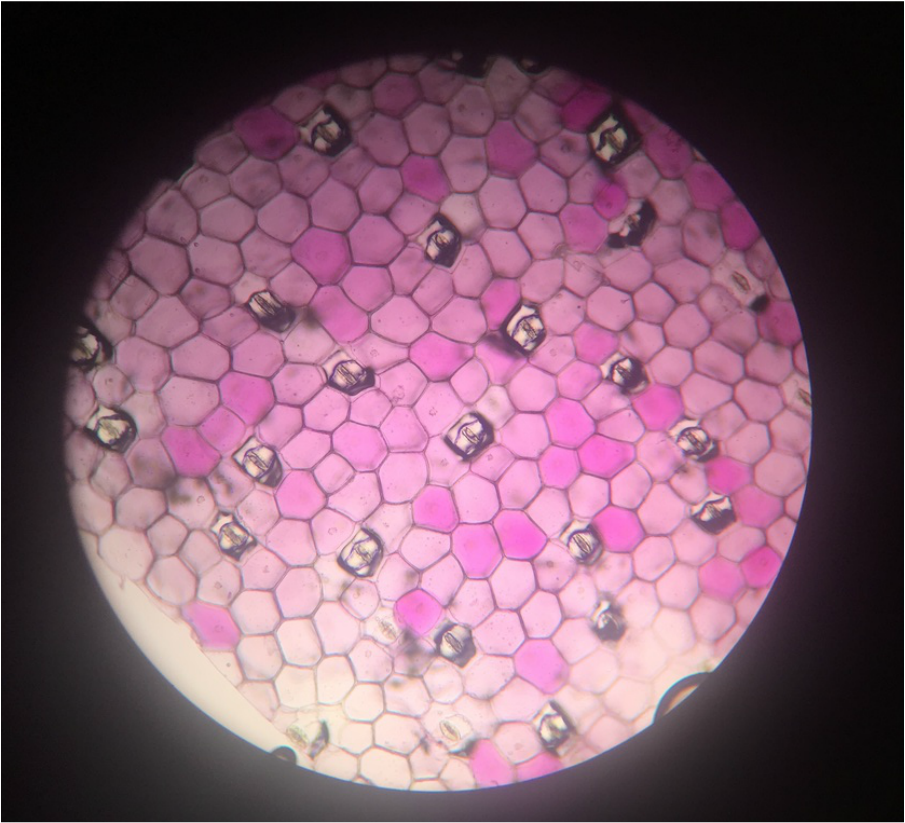
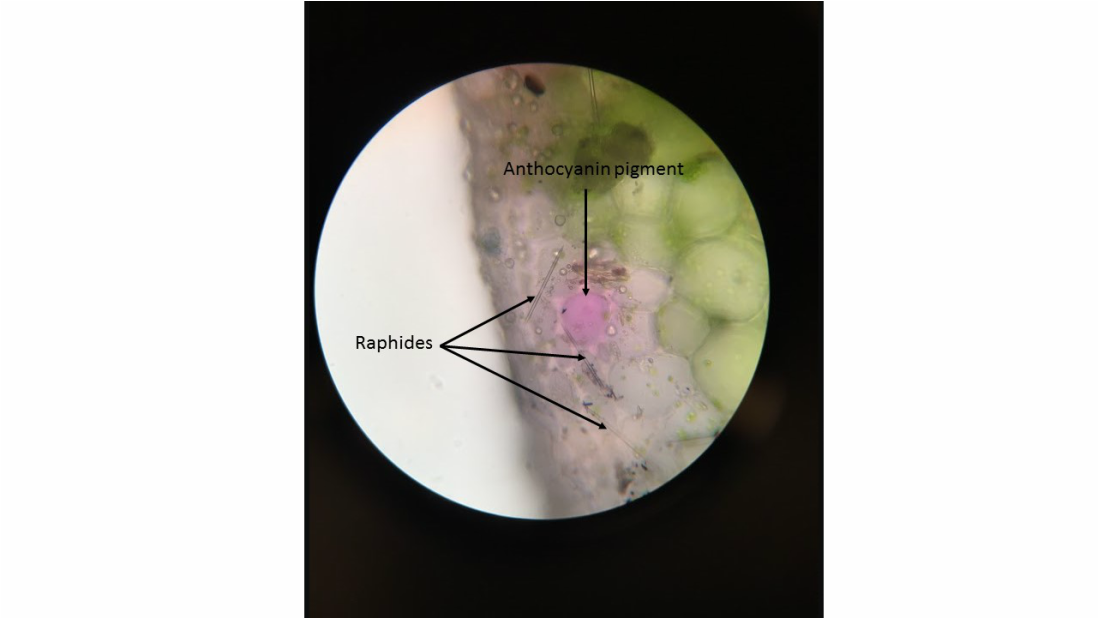
- Wide View of Lower Epidermis of Inch Plant, Tradescantia zebrina , Showing Presence of Guard Cells in Stomatal Complexes- Sample Not Stained - Epidermal Peel Section - Total Magnification 100X - Slide Prepared by Kelsey Hymel - Photo Credit: Kelsey Hymel ***NOTE: Also, the colour is purple from the presence of anthocyanin pigments in the vacuoles.***
LAB FIVE:
GMO Investigation Part One
This week we learned the difference between biotechnology- or the science of enhancing natural plants with scientific methods such as hydroponics [growing plants without soil]- and genetic engineering- or the science of physically changing the molecular and genetic composition of a known organism to create a new modified organism which could not be found in nature on its own.
We watched a documentary called "The Future of Food" which made a compelling case that GMO (genetically modified organisms) are spreading globally, and the average consumer cannot be assured that what they are eating off the grocery shelves is not GMO. Thus, being eager scientists in search of experimental proof, we brought in food samples from which to extract DNA, run PCR (polymerase chain reaction), and be able to see for ourselves if GMO food is right in our own houses without our knowledge.
We watched a documentary called "The Future of Food" which made a compelling case that GMO (genetically modified organisms) are spreading globally, and the average consumer cannot be assured that what they are eating off the grocery shelves is not GMO. Thus, being eager scientists in search of experimental proof, we brought in food samples from which to extract DNA, run PCR (polymerase chain reaction), and be able to see for ourselves if GMO food is right in our own houses without our knowledge.

We chose "Simply Organic Scoops" by Tostitos as our test food. We hypothesize that these chips will not have GMO ingredients because they are organic which is supposed to mean we can trust them to be pure and free of chemical influence. INGREDIENTS:
Organic yellow corn, organic expeller pressed Sunflower oil, and sea salt. - Photo Credit: Kelsey Hymel
Step One: Extraction of DNA from Food Samples
->The screw cape tubes that we pipetted the DNA containing slurries into were pre-loaded by Dr. LP with "InstaGene Matrix". This matrix has negatively charged microscopic beads that grab the metal ions out of our slurries. Without the Magnesium ions, the naturally present enzymes released from the cellular contents cannot perform their breakdown reactions, and the DNA is saved from being denatured.<-
Step Two: Setting Up Our PCR Reactions
We prepared to run two PCR reactions on each of our three DNA samples (Non-GMO -, Tostito's, and GMO + Control). In one PCR preparation we mixed each type of DNA with PMM or Plant Master Mix [bright green] which has primers which will locate a DNA sequence that is common in all plants [chloroplast from photosystem II], and this PCR will show whether we successfully extracted plant DNA from our Tostito's food sample. In the other PCR preparation we mixed each type of DNA with GMM or GMO Master Mix [bright pink] which has primers that will locate DNA sequence [CaMV 35S or NOS] that is common in most (85%) of all Genetically Modified Organisms (Plants).
Watch This Space for Our Exciting Results!
Will our Tostito's "Simply Organic Scoops" be shown to contain Genetically Modified Organisms?!
LAB SIX:
GMO Investigation Part Two
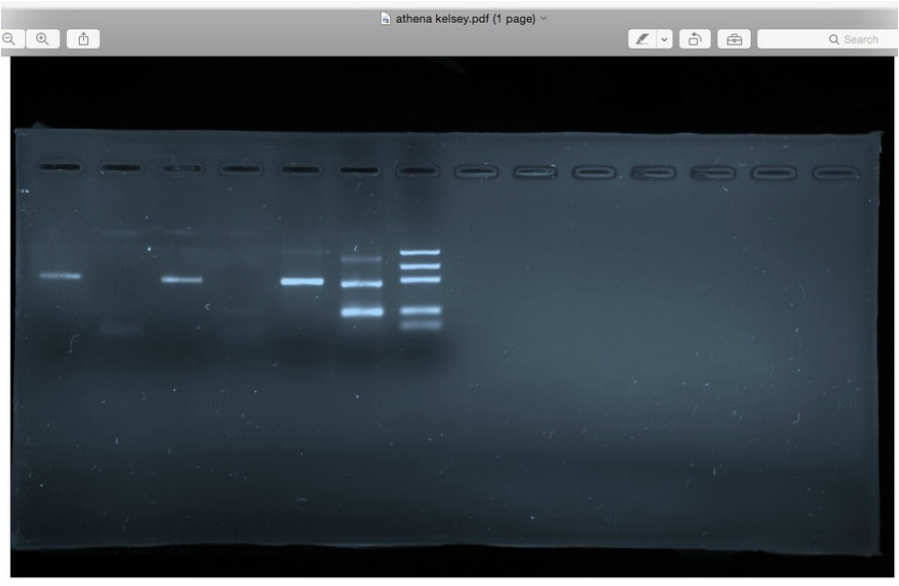
This week we used our PCR products to run gel electrophoresis in order to visualize the results of our PCR amplification from last week. We used both positive and negative controls in order to ensure that we did in fact extract DNA and then have both a GMO and non-GMO result to compare our food sample against.
First we mixed our extracted DNA samples with Orange G dye which helped to keep of DNA in solution and also contained tracking dye of known molecular size so that we can tell when the gel electrophoresis has run to completion. As DNA is negatively charged it runs down the gel to the positively charged end. Also, in lane seven of our gel, we loaded a molecular mass ruler in order to make reading our result more accurate.
We used 2% agarose gel with 30 minutes of run time at 100 volts.
First we mixed our extracted DNA samples with Orange G dye which helped to keep of DNA in solution and also contained tracking dye of known molecular size so that we can tell when the gel electrophoresis has run to completion. As DNA is negatively charged it runs down the gel to the positively charged end. Also, in lane seven of our gel, we loaded a molecular mass ruler in order to make reading our result more accurate.
We used 2% agarose gel with 30 minutes of run time at 100 volts.
RESULT: USDA CERTIFIED ORGANIC TOSTITOS SHOWED GMO POSITIVE RESULT!
OF COURSE, TO BE DULY DILIGENT WE WOULD WANT TO RETEST OUR SAMPLE TO CONFIRM; BUT THIS HI-LIGHTS THE NEED FOR GMO LABELING. WE PURCHASED "USDA ORGANIC" CHIPS TRYING TO AVOID CHEMICALS / ARTIFICIAL ELEMENTS, AND- WHILE PAYING MORE FOR LESS VOLUME-IN THE END WE WERE STILL INGESTING GENETICALLY MODIFIED ORGANISMS.
LAB SEVEN:
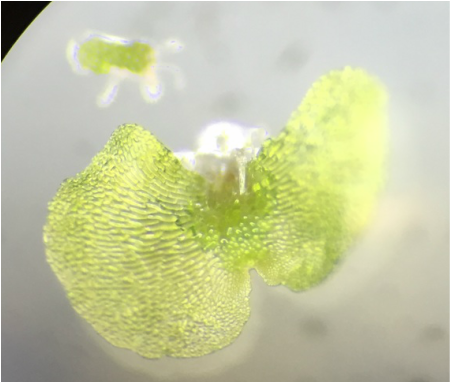
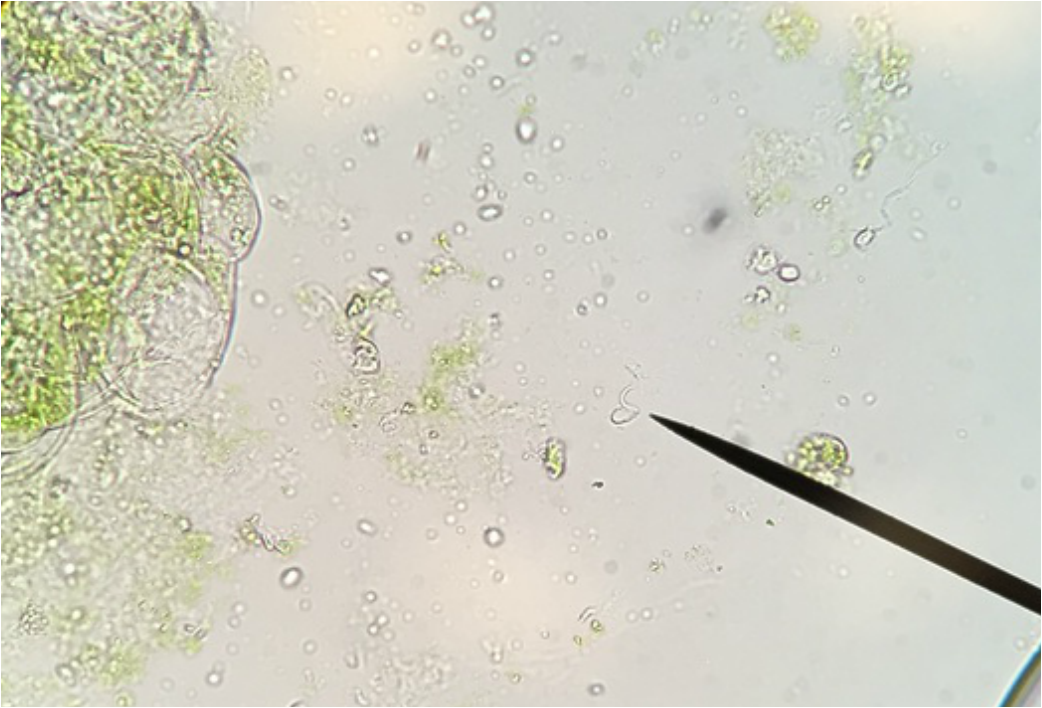
Bryophytes (Non-Vascular Plants) and Ferns (Seed- Free Plants)
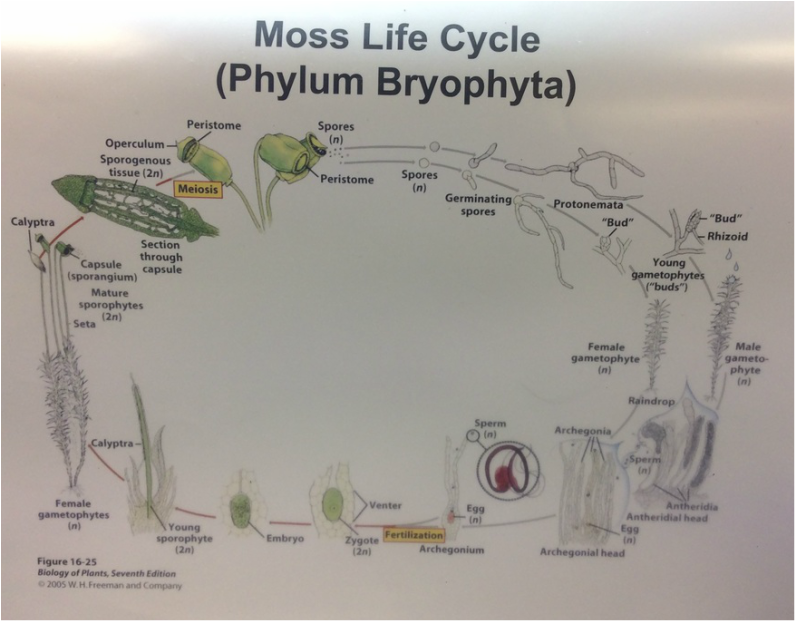
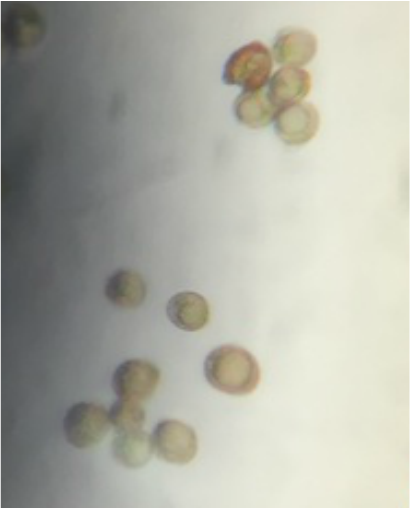
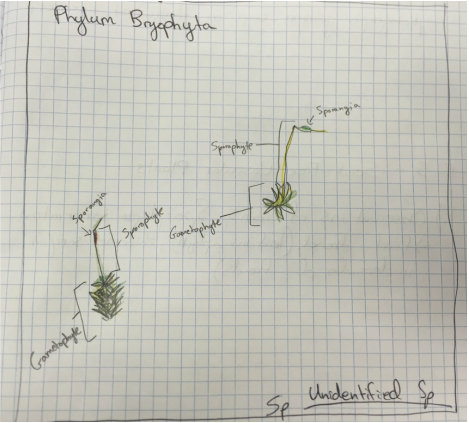
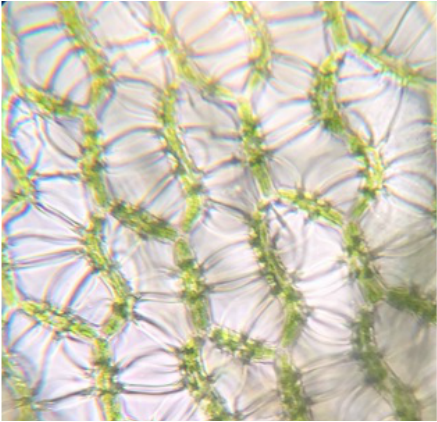
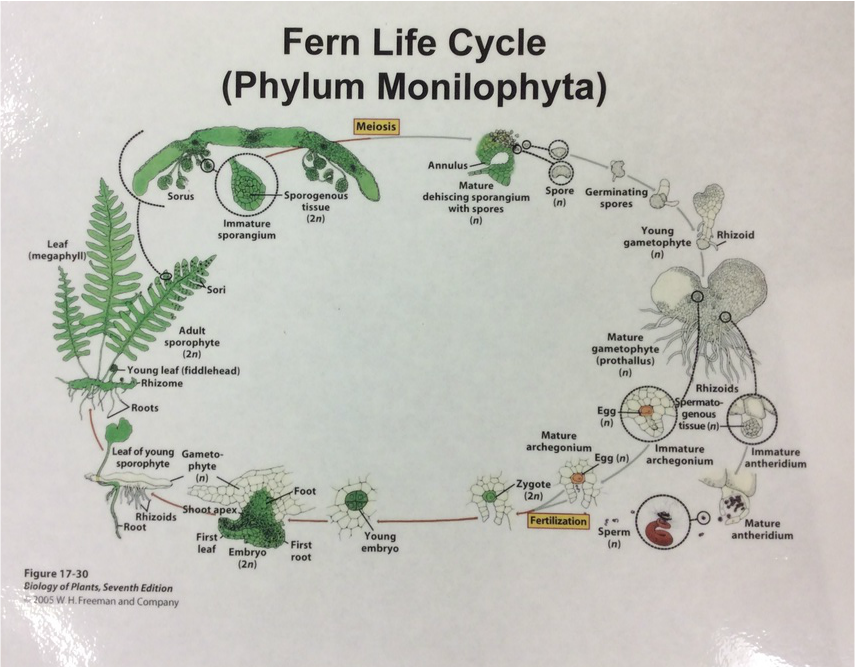
This week in lab we got up close and personal with representatives of Phylum Bryophyta (true mosses), Phylum Hepaticophyta (liverworts) and Phylum Monilophyta (ferns). We learned about their specialized life cycles and observed the gametophyte and sporophyte generations of each taxon.
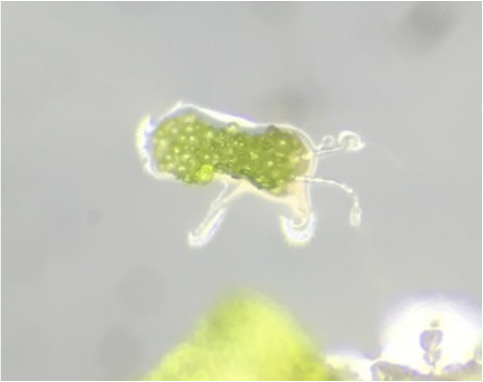
"My favourite part of this lab was getting to see the male and hermaphrodite gametophytes. Marvellous, microscopic masterpieces of biological ingenuity. Fantastic." - Athena Downes
"My favorite part of the lab was the examination of the leaf-like structure of Sphagnum sp. (or "peat moss") since it truly confirmed that mosses lack vascular tissue. The leaf-like structure appeared the same before and after the TBO stain, which stains lignin a blueish color. Though the leaf-like structure does resemble a leaf, this test proves that it is in fact NOT a true leaf as it lacks vascular tissue that would've been visible with the TBO stain if present." - Kelsey Hymel
"My favourite part of this lab was getting to see the male and hermaphrodite gametophytes. Marvellous, microscopic masterpieces of biological ingenuity. Fantastic." - Athena Downes
"My favorite part of the lab was the examination of the leaf-like structure of Sphagnum sp. (or "peat moss") since it truly confirmed that mosses lack vascular tissue. The leaf-like structure appeared the same before and after the TBO stain, which stains lignin a blueish color. Though the leaf-like structure does resemble a leaf, this test proves that it is in fact NOT a true leaf as it lacks vascular tissue that would've been visible with the TBO stain if present." - Kelsey Hymel
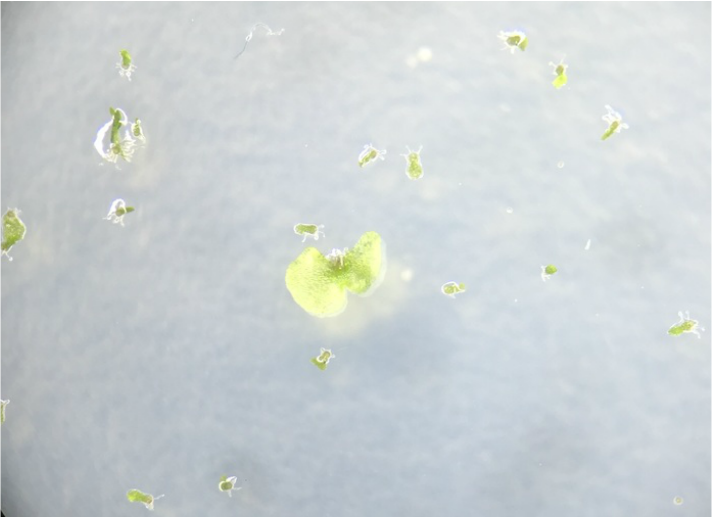
-the Mosses:
-the Liverwort:
(Phylum Hepaticophyta)
-the Ferns:
LAB EIGHT:
Gymnosperms and Angiosperms (Seed Plants)
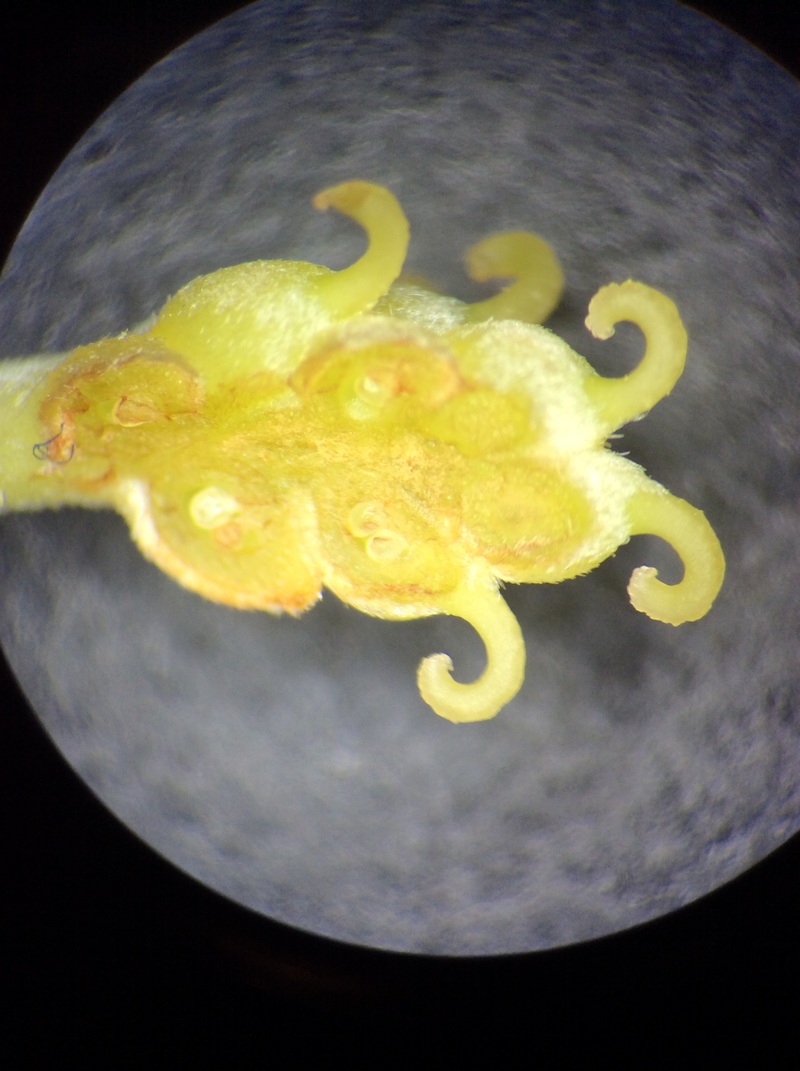
This week we took a campus walk to explore Armstrong's on-site arboretum and experienced for ourselves some living representatives of gymnosperms and angiosperms. As we visited each specimen we talked about the life cycles and sequences of events for each taxon. For gymnosperms we visited a cycad, a ginkgo, a gnetophyte, and a conifer; and then for angiosperms we collected flower samples and dissected them back in the lab.
"My favorite specimen in this lab was definitely getting to see the ginkgo tree that is here on campus! I did not know that we had one, and I really enjoyed the unique opportunity to see a such a special species firsthand!." -Athena
"My favorite specimen in this lab was the Dawn Redwood. I loved hearing the story of how scientists only knew of this plant species from fossilized remains and thought to be extinct until living members were found only several decades ago. The fact that this tree resides on campus shows the power of conservation as Dr. LP told our class that once this species was found, its seeds were shipped to campuses all over the United States. It's really neat to know that I pass by a "living fossil" of sorts nearly everyday." -Kelsey
"My favorite specimen in this lab was definitely getting to see the ginkgo tree that is here on campus! I did not know that we had one, and I really enjoyed the unique opportunity to see a such a special species firsthand!." -Athena
"My favorite specimen in this lab was the Dawn Redwood. I loved hearing the story of how scientists only knew of this plant species from fossilized remains and thought to be extinct until living members were found only several decades ago. The fact that this tree resides on campus shows the power of conservation as Dr. LP told our class that once this species was found, its seeds were shipped to campuses all over the United States. It's really neat to know that I pass by a "living fossil" of sorts nearly everyday." -Kelsey
LAB NINE:
Root and C-Fern Observations (& Shoot Cuttings)
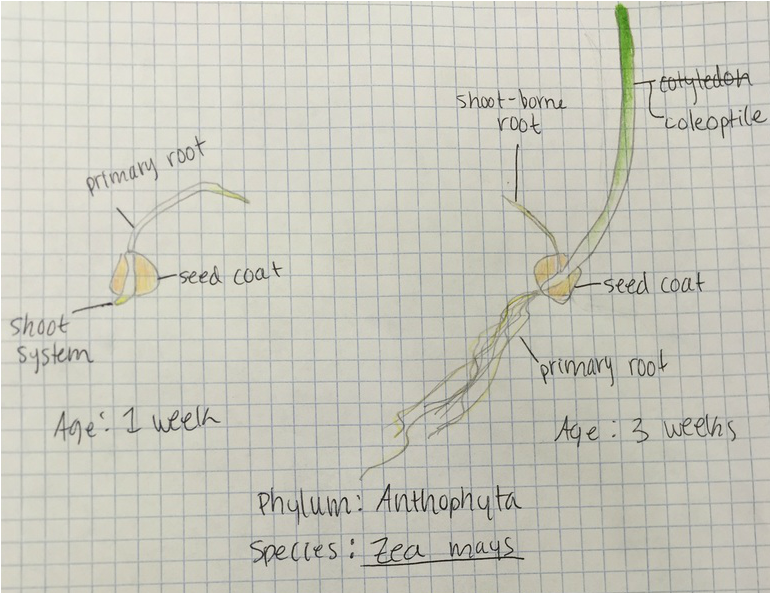
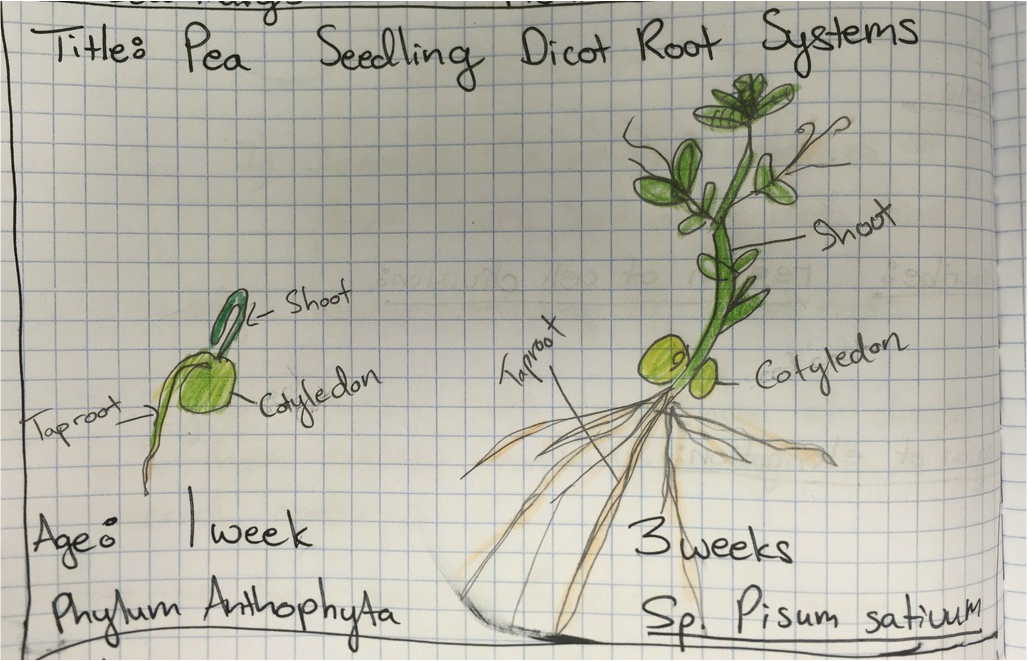
This week in lab we used our sporophyte agar plates and sketches of monocot and dicot roots to identify the external features of plant roots, including root hairs. We also examined the internal structures with our compound microscopes and cross sections of the one week old monocot and dicot roots. We learned to distinguish the external morphology and internal anatomy of dicot roots from monocot roots.
Roots anchor plants to substrate; absorb water and minerals; conduct water, minerals, and carbohydrates; and store carbohydrates in the form of amyloplast starches.
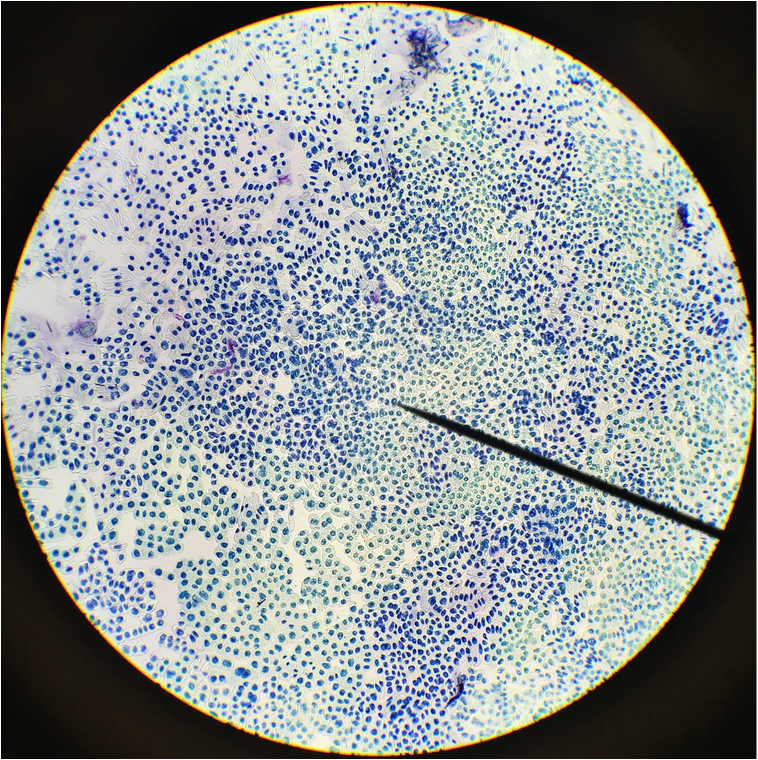
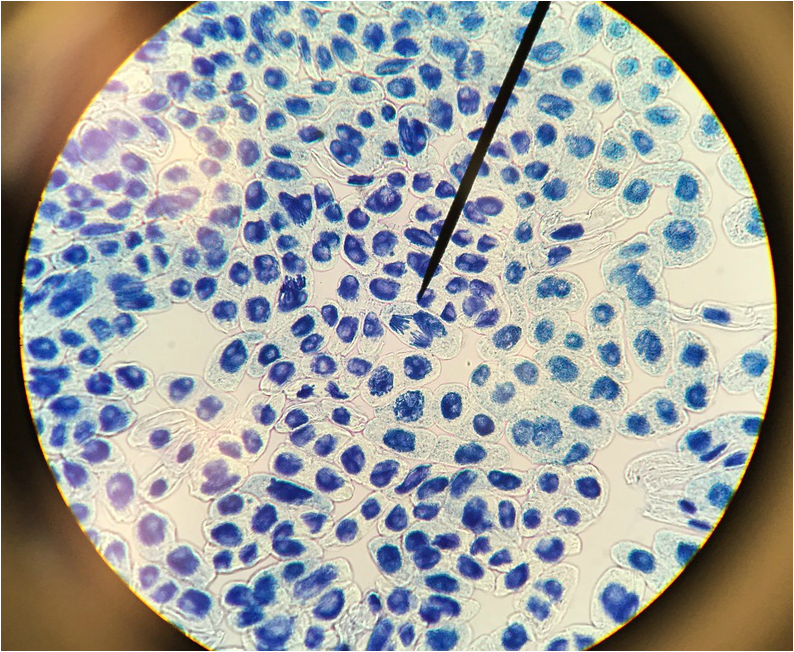
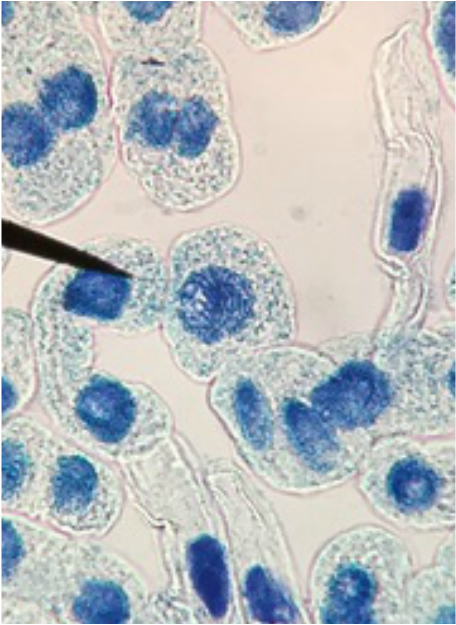
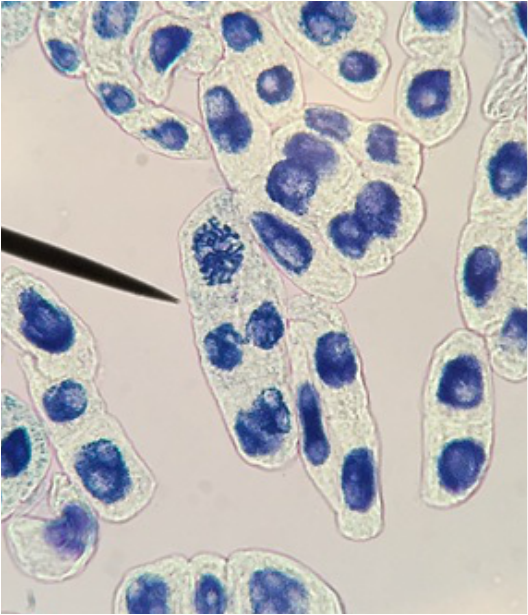
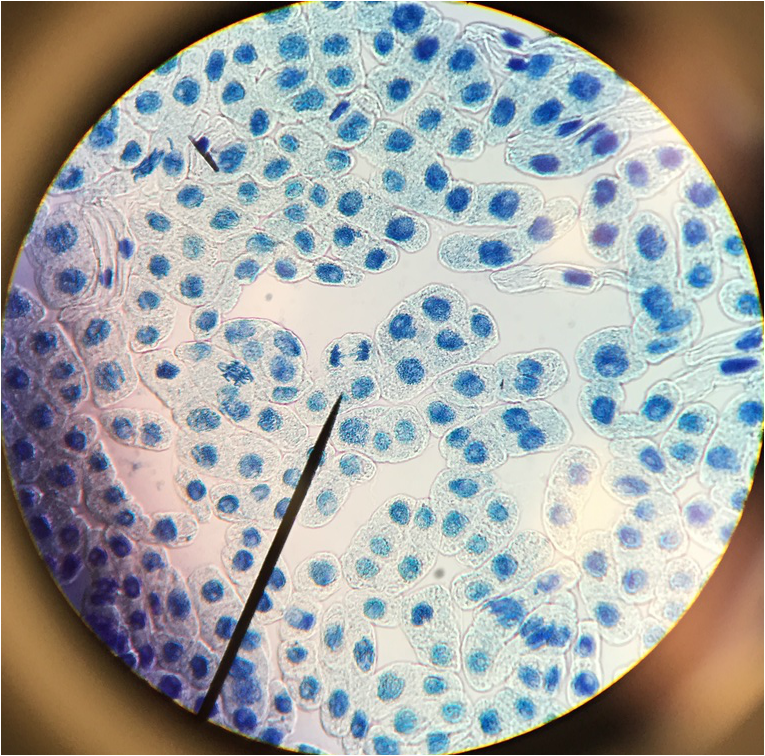
"My favourite thing this lab period was the mitotic squash. What a joy to see the stages of mitosis frozen in time, and to think about how this is the way that living things grow. Amazing!" -Athena Downes
"My favorite part of this lab was the primary root cross-section I prepared from the monocot Zea mays (or corn). Generally, I am able to prepare my own slides with little trouble, however, this time that was not the case. Not only did I try using the stain for suberin unsuccessfully, I had to prepare two additional slides using the TBO stain, which stains for lignin, in order to slice the cross-section just right. The difficulty of cutting the primary root from such a small specimen proved to be a challenge, and though I do enjoy a challenge, I was not alone in the frustration of producing an adequate cross-section. However, patience and consistency eventually paid off, and I was pleased with my final results at last!" -Kelsey Hymel
Roots anchor plants to substrate; absorb water and minerals; conduct water, minerals, and carbohydrates; and store carbohydrates in the form of amyloplast starches.
"My favourite thing this lab period was the mitotic squash. What a joy to see the stages of mitosis frozen in time, and to think about how this is the way that living things grow. Amazing!" -Athena Downes
"My favorite part of this lab was the primary root cross-section I prepared from the monocot Zea mays (or corn). Generally, I am able to prepare my own slides with little trouble, however, this time that was not the case. Not only did I try using the stain for suberin unsuccessfully, I had to prepare two additional slides using the TBO stain, which stains for lignin, in order to slice the cross-section just right. The difficulty of cutting the primary root from such a small specimen proved to be a challenge, and though I do enjoy a challenge, I was not alone in the frustration of producing an adequate cross-section. However, patience and consistency eventually paid off, and I was pleased with my final results at last!" -Kelsey Hymel
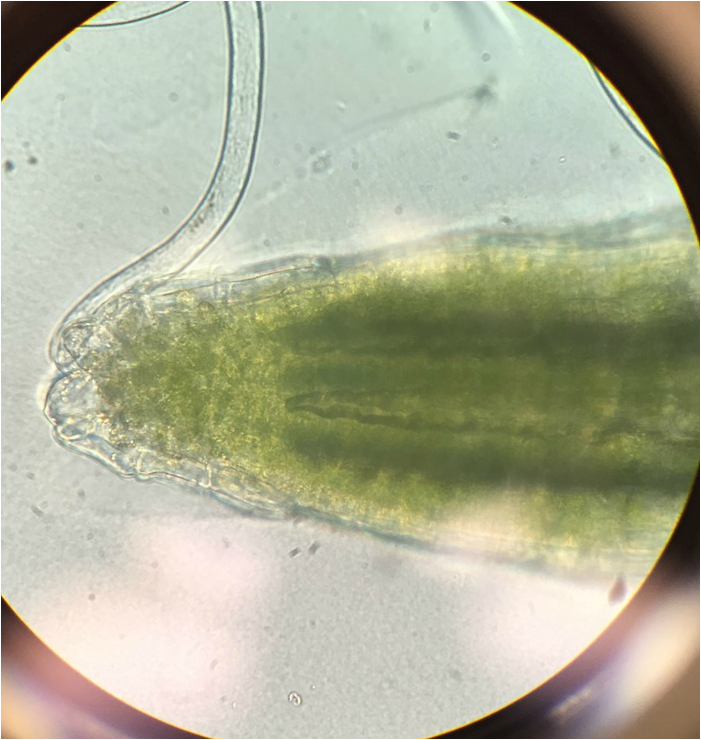
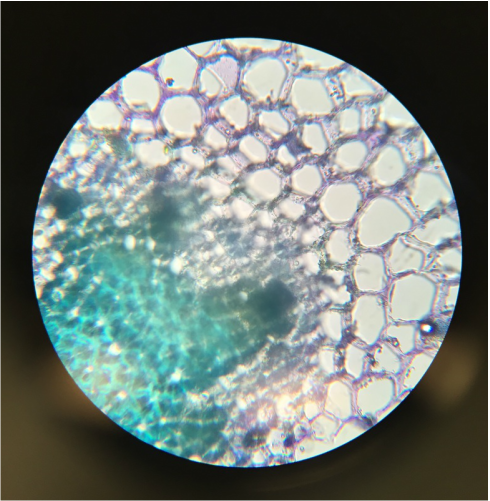
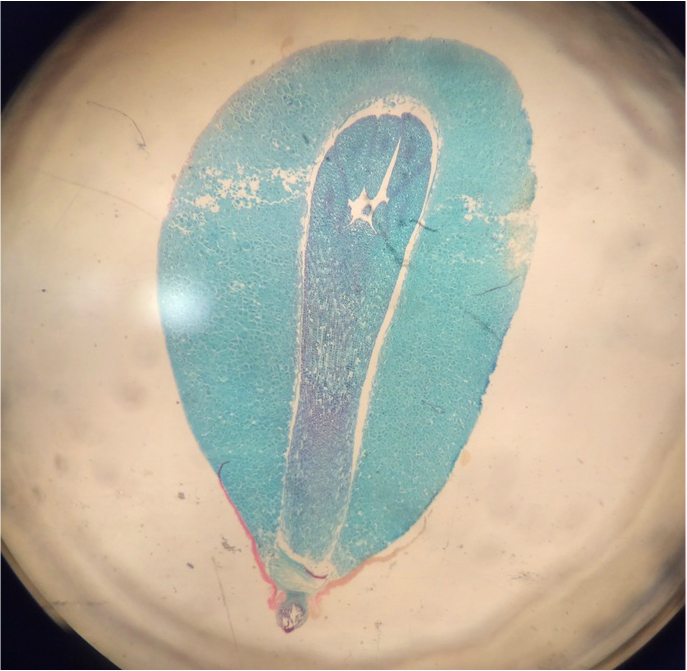
Observation of C-Fern Roots (Ceratopteris richardii) - Wholemount Slide Prepared by Athena Downes - Photo credit: Athena Downes - Compound Microscope - Total Magnification: 400X - Notice the visible root cap/tip just behind the primary root. Also the three areas of growth: the region of cell division, the region of elongation, and the region of maturation. -
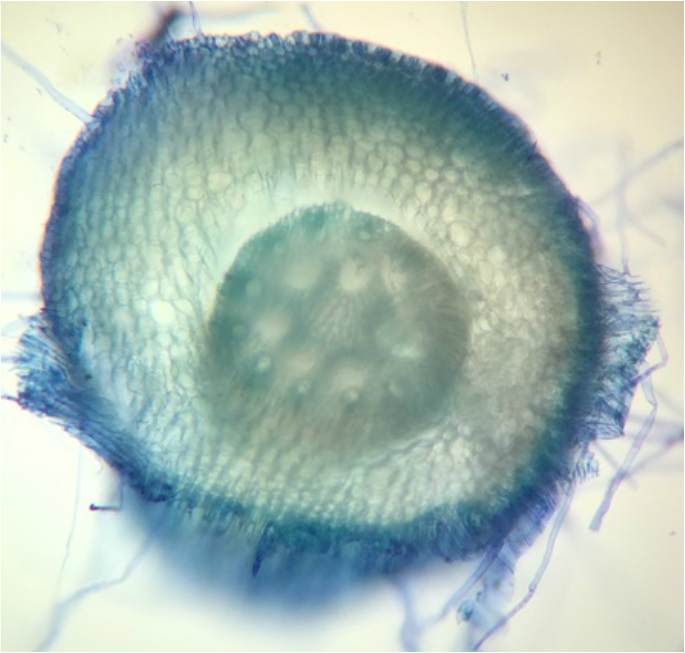
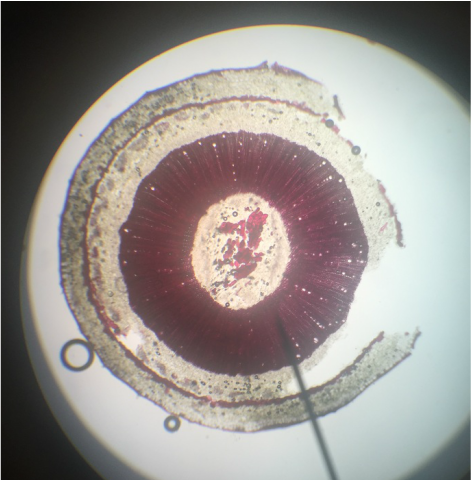
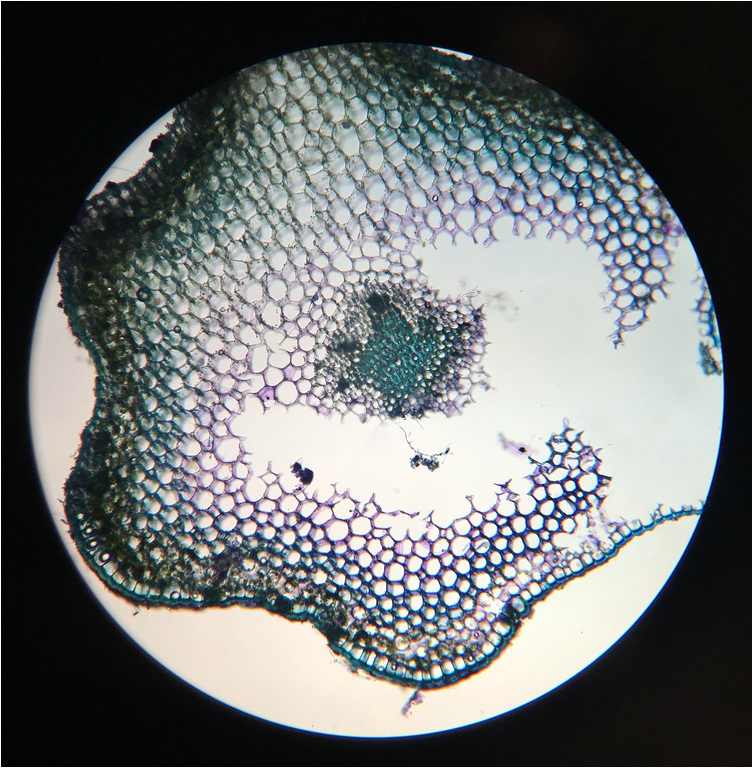
Monocot Corn ( Zea Mays ) Cross Section of Primary Root - Age 1 Week - Slide Prepared by Kelsey Hymel - Photo Credit: Kelsey Hymel - TBO Stain Applied Showing Blue-Green Color of Lignin Containing Cell Walls - Also Note the Parenchyma Cells in the Center Core, The Large Circular Xylem, and the Smaller Circular Phloem -
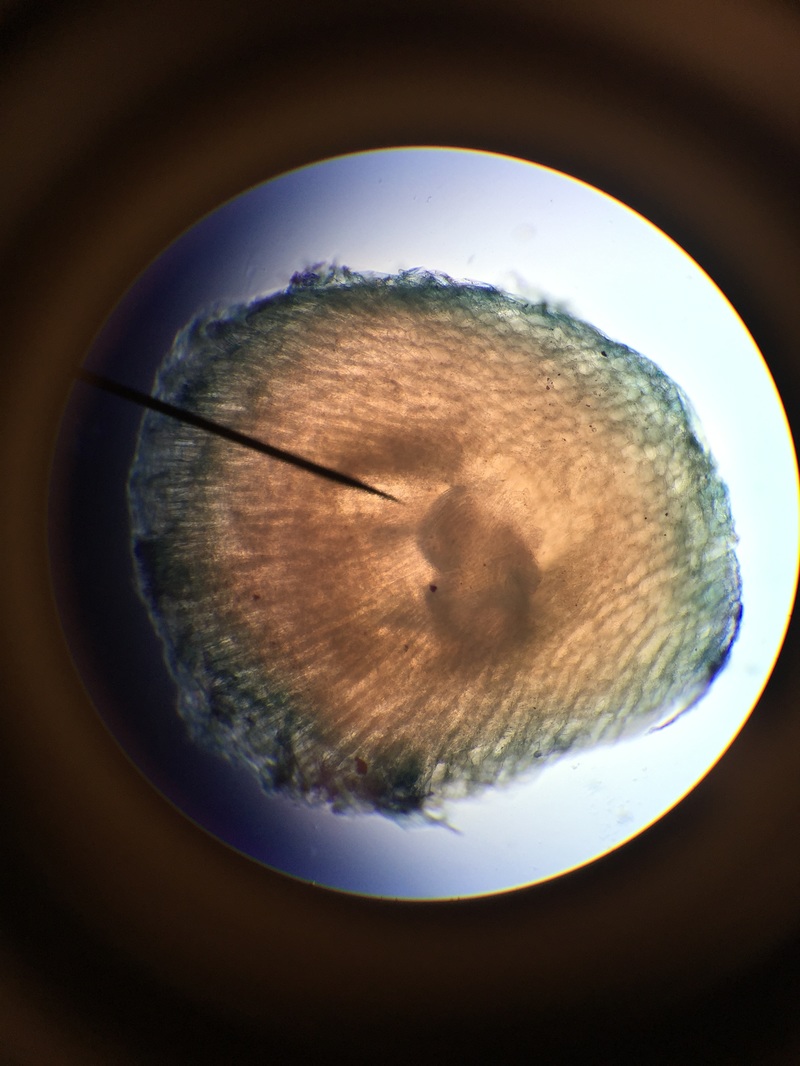
Dicot Pea (Pisum sativum) Cross Section of Primary Root - Age 1 Week - Slide Prepared by Athena Downes - Photo Credit: Athena Downes - TBO Stain Applied to Highlight in Blue-Green Presence of Lignin Containing Tissues - Compound Microscope - Total Magnification: 400X - Note the Outer Epidermis, The Center Should Reveal a Cross-Shaped Group Of Vascular Tissues with Both Xylem and Phloem, but the Double Stain has Obscured These Tissues -